Which of the following could exist as a cistrans isomer? a. CH 3 CH 2 CH 2
Question:
Which of the following could exist as a cis–trans isomer?
a. CH3CH2CH2CH3
b.
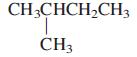
c.
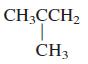
d. CH3CHCHCH3
e. none of these
Transcribed Image Text:
CH;CHCH,CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds show cis-trans isomerism? Draw the cis and trans isomers of the ones that do. (a) CH3CH=CHCH3 (b) CH3-C¡C-CH3 (c) CH2=C(CH3)2 (d) (e) (f) CH3-CH=N-CH3...
-
a. Which of the following compounds can exist as cis-trans isomers? b. For those compounds, draw and label the cis and trans isomers. 1. CH3CH==CHCH2CH3 2. 3. CH3CH2CH==CH3 4. CH3CH2CH==CH2 ,
-
Which of the following compounds can exist as cis-trans isomers? Draw their structures. a. Pentene b. 3-heptene c. 2-methyl-2-pentene d. 2-hexene
-
Calculate the following integral approximately by using the Simpson's method: 3.6 f. f(x) dx X 0.0 0.6 1.2 1.8 2.4 3.0 3.6 f(x) 0 0.13 0.05 0.6 0.43 0.74 0.93
-
1. If a fixed number c is added to all measurements in a data set, the deviations (x - ) remain unchanged (see Exercise 2.52). Consequently, s2 and s remain unchanged. 2. If all measurements in a...
-
The following information was obtained from the ledger of Stillwell Emporium, Inc., at the end of 2016: Stillwell analyzes its operating expenses at the end of each period in order to prepare an...
-
Question: Police bang down the door of Mary Beth's apartment, enter without her permission, and search the apartment. They had no warrant. The officers discover a machine used to manufacture fake...
-
Amanda M is a regional manufacturer and wholesaler of high-quality chocolate candies. The companys sales and collection process is as follows. Amanda M makes use of an enterprise-wide information...
-
11. Recently you have received a tip that the stock of Bubbly Incorporated is going to rise from $57 to $61 per share over the next year. You know that the annual return on the S&P 500 has been 9....
-
Consider the process model in Fig. 7.14. This model captures a simplified process for handling applications for mortgages. There are two checks involved. CT1 deals with a check of the financial...
-
How many isomers exist that have the formula C 4 H 8 ? a. Two b. Three c. Four d. Five e. Six
-
The correct formula for butane is a. C 2 H 6 b. C 8 H 18 c. CH 4 d. C 4 H 10 e. C 3 H 6
-
Thermal transfer printing transfers ink from a ribbon onto paper through a combination of heat and pressure. Different types of printers use different sizes of ribbons. A ribbon manufacturer has...
-
Hudson Company reports the following contribution margin income statement. HUDSON COMPANY Contribution Margin Income Statement For Year Ended December 31 Sales (10,700 units at $300 each) Variable...
-
Situation 1 2 3 4 Lease term (years) Asset's fair value be exercised? 4 4 4 4 Asset's useful life (years) 6 $44,000 Purchase option that is reasonably certain to No Annual lease payments Beginning of...
-
A manufacturing company operates two processes. Output from Process 1 is transferred as input to Process 2. Output from Process 2 is the finished product. Data for the two processes in January are as...
-
Coffer Company is analyzing two potential investments. Project X Project Y Cost of machine $ 85,470 $ 65,000 Net cash flow: Year 1 33,000 3,000 Year 2 33,000 30,000 Year 3 33,000 30,000 Year 4 0...
-
1. The following information was taken from the record of Anton Manufacturing Inc.: Increase in finished goods, P100,000; Increase in work in process, P110,000; Decrease in raw materials P96,000;...
-
Richard Penn lives in Harrisburg, Pennsylvania. Richard is the president of an architectural firm. Richard has become known throughout the community for excellent work and honesty in his business...
-
Chapter 9 Stock Valuation at Ragan Engines Input area: Shares owned by each sibling Ragan EPS Dividend to each sibling Ragan ROE Ragan required return Blue Ribband Motors Corp. Bon Voyage Marine,...
-
A 25 kg piston is above a gas in a long vertical cylinder. Now the piston is released from rest and accelerates up in the cylinder reaching the end 5 m higher at a velocity of 25 m/s. The gas...
-
The rolling resistance of a car depends on its weight as: F = 0.006 mg. How far will a car of 1200 kg roll if the gear is put in neutral when it drives at 90 km/h on a level road without air...
-
A mass of 5 kg is tied to an elastic cord, 5 m long, and dropped from a tall bridge. Assume the cord, once straight, acts as a spring with k = 100 N/m. Find the velocity of the mass when the cord is...
-
A main objective of employment legislation in Canada is to prevent employers from exploiting paid workers. True False * 1 point Human rights legislation: * 1 point primarily affects compensation and...
-
Question 3 (0.5 points) Saved Within the DMAIC improvement methodology, which step would be most concerned with understanding the existing process capability and collecting in-process data? A) Define...
-
Given this code: SELECT product_name, list_price, CASE WHEN list price> 0 AND list_price 600 THEN 'Mass' WHEN list_price >= 600 AND list_price < 1000 THEN 'Economy' WHEN list price >= 1000 AND...

Study smarter with the SolutionInn App



