Would ethanol (CH 3 CH 2 OH) be a suitable solvent in which to perform the following
Question:
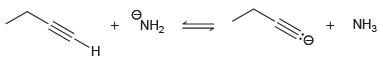
Transcribed Image Text:
ONH2 + NH3 NH2 H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
No because the leveling effect ...View the full answer

Answered By

Jacob Festus
I am a professional Statistician and Project Research writer. I am looking forward to getting mostly statistical work including data management that is analysis, data entry using all the statistical software’s such as R Gui, R Studio, SPSS, STATA, and excel. I also have excellent knowledge of research and essay writing. I have previously worked in other Freelancing sites such as Uvocorp, Essay shark, Bluecorp and finally, decided to join the solution inn team to continue with my explicit work of helping dear clients and students achieve their Academic dreams. I deliver, quality and exceptional projects on time and capable of working under high pressure.
4.90+
1260+ Reviews
2853+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Sciences questions
-
Suggest suitable reagents to perform the following transformation: Racemic
-
Design an op amp circuit to perform the following operation: Vo = 3v1 - 2v2 All resistance must be ( 100 k(.
-
Natural gas containing a mixture of methane ethane propane and butane is burned in a furnace with excess air. (a) One hundred k mol/h of a gas containing 94.4 mole% methane, 3.40% ethane 0.60%...
-
Peter, Emma, and Kyler played chess with each other. Peter won 4 games and lost 2 games. Emma won 3 games and lost 3 games. If Kyler lost 3 games, how many games did he win?
-
1. Download and read the Kaplan and Norton (2004b) article "How Strategy Maps Frame an Organization's Objectives" 2. Download and read the Ingham (2011) article "Using a Human Capital Scorecard as a...
-
Use the concept of volume to explain way the determinant of a 3 ( 3 matrix A is zero if and only if A is not invertible. Do not appeal to Theorem 4 in Section 3.2. [Think about the columns of A.]
-
Under the double declining-balance method, the salvage value is ignored in the calculations; however, the asset is not depreciated below the salvage value. (True/False)
-
The enrollments of the 13 public universities in the state of Ohio are listed below. College Enrollment University of Akron......... 26,666 Bowling Green State University.... 17,298 Central State...
-
ssets Current assets $ 32,200 Property and equipment, net 89,000 $ 121,200 Total assets iabilities and Equity Liabilities $ 36,500 Stockholders' equity: $0.70 cumulative preferred stock, $5 par, 800...
-
Robby Beamon has recently been appointed vice-president of sales and marketing for Lori's Unpublished Books Limited, a public company. Robby has come to you for advice regarding the tax implications...
-
Identify the reagents you would use to convert 2-bromo-2-methylbutane into 3-methyl-1-butyne.
-
Identify the reagents you would use to convert 1-pentene into a geminal dibromide (geminal indicates that both bromine atoms are connected to the same carbon atom).
-
A certain memory has a capacity of 8K 16. How many bits are in each word? How many words are being stored? How many memory cells does this memory contain?
-
Where on the Estate Agents legislation is the currency located? What is the date of the current version of this legislation?
-
What are the social needs and the importance in today's business world? Explain in details.
-
The accountant of JYD Corporation prepared the following cost analysis report on direct labor costs for the jobs completed during the previous months: JobActual Hrs. at Actual RatesActual Hrs. at...
-
Collingwood Homes has a bond issue outstanding that pays an 8.5 percent coupon and has a yield to maturity of 9.16%. The bonds have a par value of $1,000 and a market price of $944.30. Interest is...
-
Where do you see commercial opportunity to grow The UK's fastest-growing management consultancy, over the next a year and a half?
-
Location pooling is most effective at generating which of the following changes to performance objectives? a. Increasing the gross margin. b. Increasing the target in-stock probability. c. Decreasing...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Heating ethanol with sulfuric acid is one method used for the preparation of diethyl ether. Show all of the steps in the mechanism for this reaction: H,SO4 2 CH;CH,OH CH;CH,-0-CH,CH, + H2O
-
When an aqueous solution of (R)-2-butanol is treated with a catalytic amount of sulfuric acid, slow racemization of the alcohol occurs. Show all of the steps in the mechanism for this process.
-
Show all of the steps in the mechanism and explain the stereo chemistry for this reaction: Br , Br + . + C-C CH3 . CH3 .
-
The best approach for determining the Wednesdays on which food orders were received is to put the ReceivingSlip table in a new query, populate the query with the needed attributes, and: 1) Create the...
-
Cullumber Corporation is a diversified company that operates in five different industries: A, B, C, D, and E. The following information relating to each segment is available for 2021. A B C D E Sales...
-
Make-or-Buy Decision Matchless Computer Company has been purchasing carrying cases for its portable computers at a purchase price of $57 per unit. The company, which is currently operating below full...

Study smarter with the SolutionInn App


