Would you expect the following compound to be aromatic? Justify your answer. OR N-
Question:
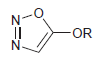
Transcribed Image Text:
OR N-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
Yes The lone pairs on the nitrogen atoms do not cont...View the full answer

Answered By

Shristi Singh
A freshman year metallurgy and material science student in India.
4.80+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Would you expect the following compound to be aromatic? Explain your answer.
-
How many absorptions would you expect the following compound to have in its 1H and 13C NMRspectra?
-
Do you expect the following compound to be chiral? Explain your answer (consider whether this compound is super-imposable on its mirror image).
-
Solve each system. x + 4y = - 3x + 2y = 17 -9
-
Discuss the role culture plays in organizational change. Include the following: Define organizational culture. Discuss the roots of organizational culture. Discuss the role organizational culture...
-
A scanning electron micrograph of xylem vessels in a corn root shows the vessels magnified by a factor of 600. In the micrograph the xylem vessel is 3.0 cm in diameter. (a) What is the diameter of...
-
During 2015, Contender Oil Company obtained the following leases: After obtaining these leases, Contender Oil Company incurred shooting rights on Leases A and C at $0.50 an acre and incurred the...
-
The demand curve for product X is given by Qdx = 460 = 4Px. a. Find the inverse demand curve. b. How much consumer surplus do consumers receive when Px = $35? c. How much consumer surplus do...
-
Bardi Trucking Co., located in Cleveland, Ohio, makes deliveries in the Great Lakes region, the Southeast, and the Northeast Am Bardi, the president, is studying the relationship between the distance...
-
As someone who came of age in the "post email" world of blogs, wikis, social networks, and other Web 2.0 technologies, you were rather disappointed to find your new employer solidly stuck in the age...
-
Do you expect the following dianion to exhibit aromatic stabilization? Explain.
-
Diphenylmethane exhibits two aromatic rings, which achieve coplanarity in the highest energy conformation. Explain. Diphenylmethane
-
The logistic growth function models the percentage, P(x), of Americans who are x years old with some coronary heart disease. Use the function to solve Exercises 4346. What percentage of 20-year-olds...
-
Show the AVL tree resulting from deleting 5 from the AVL tree below. 2 4 1 3 5 6 8 7 9
-
The partnership of Matteson, Richton, and O'Toole has existed for a number of years. At the present time, the partners have the following capital balances and profit and loss sharing percentages:...
-
Example 5.3 a) Calculate all the forces acting on each of the three members, i.e. b) The members have the following weights: Post-1400lb, boom-1500lb, brace-900lb. 1. Reaction Forces, H, v, & B. 2....
-
Consider a portable 20 cm 2 active area fuel cell operating steadily at 0.75V and 0.6 A/cm 2 The fuel utilization efficiency is 50%, and cathode stoichiometry is 2.3 expected to run for 3 days. The...
-
Suppose the U.S. and Japan trade with each other. For simplicity, assume that the only 2 goods that are traded are phones and bikes. The figure below summarizes the impact. of trade in the United...
-
Imagine that you are building an online plagiarism checker, which allows teachers in the land of Edutopia to submit papers written by their students and check if any of those students have copied...
-
Synthesize the products by drawing out reagents and intermediates along the way. `N H. OH HO HO
-
Give the structure the organic product(s) formed when HBr reacts with each of the following alkenes in the presence of peroxides, and explain your reasoning. If more than one product is formed,...
-
Give the structures of the free radical intermediates in the peroxide-initiated reaction of HBr with each of the following alkenes. CH
-
Estimate the H values for each of the following gas-phase reactions using bond dissociation energies. H2C===CH2 + Cl2 + CI-CH2CH2-C
-
Eight years ago, SW purchased 1,900 shares of Delta stock. On May 20 of the current year, it sold these shares for $38 per share. In each of the following cases, compute SWs recognized gain or loss...
-
The best approach for determining the Wednesdays on which food orders were received is to put the ReceivingSlip table in a new query, populate the query with the needed attributes, and: 1) Create the...
-
Cullumber Corporation is a diversified company that operates in five different industries: A, B, C, D, and E. The following information relating to each segment is available for 2021. A B C D E Sales...

Study smarter with the SolutionInn App


