At sufficiently high temperatures, the van der Waals equation has the form P L RT>(V m -
Question:
Figure 1.10
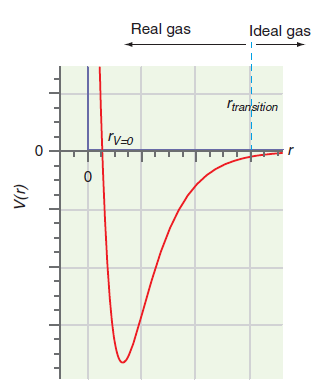
Transcribed Image Text:
Ideal gas Real gas Itrarlsition (1)A
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (16 reviews)
At high temperatures the energy of the molecule is lar...View the full answer

Answered By

Zablon Gicharu
I am an educator who possesses the requisite skills and knowledge due to interacting with students for an extended period. I provide solutions to various problems in step-by-step explanations, a well-thought approach and an understandable breakdown. My goal is to impart more straightforward methodologies and understanding to students for more remarkable achievements.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For carbon dioxide gas the constants in the van der Waals equation are = 0.364 J m 3 /mol 2 and (a) If 1.00 mol of gas at 350 K is confined to a volume of find the pressure of the gas using the...
-
In Sample Exercise 10.16, we found that one mole of Cl2 confined to 22.41 L at 0oC deviated slightly from ideal behavior. Calculate the pressure exerted by 1.00 mol Cl2 confined to a smaller volume,...
-
The parameter a in the van der Waals equation is greater for H 2 O than for He. What does this say about the difference in the form of the potential function in Figure 1.10 for the two gases? Figure...
-
A mortgage loan officer uses math on a continual basis during the mortgage lending process. When a prospective borrower applies for a loan, many calculations are made: debt-to-income- ratio,...
-
The following transactions have also occurred at Fitzgerald. 1. Options were granted on July 1, 2013, to purchase 200,000 shares at $15 per share. Although no options were exercised during fiscal...
-
Even within the new classical model, anticipated policy actions such as an increase in the money supply will affect nominal income. Explain why the adjustment of economic agents' expectations, which...
-
Windshield wipers were invented by Mary Anderson in 1903, when she observed that streetcar drivers in New York City had to open the window when it rained in order to see. Her invention consisted of a...
-
Consider the two options in the following table, both of which have random outcomes: a. Determine the expected value of each option. b. Determine the variance and standard deviation of each option....
-
E7-15 (Algo) Analyzing and Interpreting the Inventory Turnover Ratio [LO 7-5] Clark & Scott Corporation is the biggest snowmobile manufacturer in the world. It reported the following amounts in its...
-
1. Imagine that an agency or business has asked for help in gathering data about the number of sports anglers who fish off the coast of Georgia. What advice would you give about sampling? What method...
-
Define the seven aspects of customer service.
-
What are some uses for hydrogen fuel cells?
-
In a heat exchanger (see accompanying animation) a flow of hot air is cooled by a flow of water. Identify the interactions treating (a) The entire heat exchanger as the system (b) One of the streams...
-
The Regal Cycle Company manufactures three types of bicycles-a dirt bike, a mountain bike, and a racing bike. Data on sales and expenses for the past quarter follow: Sales Variable manufacturing and...
-
Molly is shopping for personal auto liability insurance for her new car. She has asked for a quote at the basic limit of $50,000 per accident and an option with a $1 million per accident limit. Molly...
-
Test the following series for convergence. Show every step you use to get the answer. (a) (b) n=1 n-2 3Thn n! (-1)+1 n(In(n))
-
Following is a list of credit customers along with their amounts owed and the days past due at December 31. Following that list are five classifications of accounts receivable and estimated bad debts...
-
25 26 27 28 Provide at least FIVE (5) examples of the specific records that you may need to keep in relation to the financial aspects of a project. What strategies can a Project Manager adopt to help...
-
In commercials for Bounty paper towels, the manufacturer claims that they are the quicker picker-upper, but are they also the stronger picker-upper? Two of Mr. Tabors statistics students, Wesley and...
-
Using the information presented in Problem 13.4B, prepare a partial statement of cash flows for the current year, showing the computation of net cash flows from operating activities using the...
-
For PbI2(s) = 0Pb+(aq) + 2 r(aq), K = 1.4 X 10-8 at 25C and the standard Gibbs energy of formation ofPbI2(s) is -173.64 k] mol ". Calculate the standard Gibbs energy of formation of PbI2 (aq).
-
Write the cell reaction and electrode half-reactions and calculate the standard emf of each the following cells: (a) Ptl C12 (g) I HCl (aq) 11 K, Cr04 (aq) IAg, Cr04(s) IAg (b) Pt 1 Fe3+(aq),Fe2+(aq)...
-
Devise cells in which the following are the reactions and calculate the standard emf in each case: (a) 2 Na(s) + 2 H20 (l) --7 2 NaOH (aq) + H2 (g) (b) H2 (g) + I2 (g) --72 HI (aq) (c) H30+ (aq) + OW...
-
On 1 January 20.1 SA Traders purchased office furniture for R80 000. The financial year-end of SA Traders is 31 December.Calculate the accumulated depreciation for 20.3 by using the reducing-balance...
-
Customs agents are on the lookout for situations where the value of the transaction (invoice value) may not represent the true value of goods being imported. Identify the 3 situations most likely to...
-
Iverson, Inc. has $1,294,000 of sales revenue, $692,000 of variable costs, and $692,000 of fixed costs in 2023. Assuming the contribution margin percentage stays the same in 2024, what will the break...

Study smarter with the SolutionInn App


