Explain the direction of the chemical shifts for Fe(0), Fe(II), and Fe(III) in Figure 22.20. Fe 2p
Question:
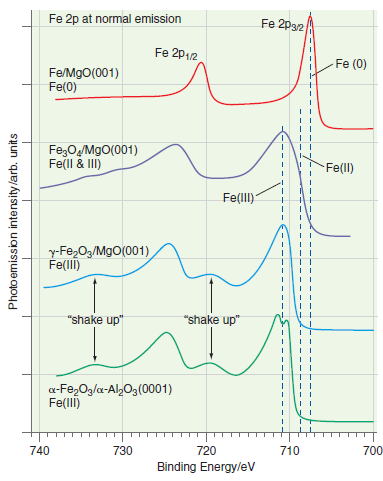
Transcribed Image Text:
Fe 2p at normal emission Fe 2p32 Fe 2p12 Fe (0) Fe/MgO(001) Fe(0) Fe;OM9O(001) Fe(ll & II) Fe(ll) Fe(lI) y-Fe,O3/MgO(001) Fe(llI) "shake up" "shake up" a-Fe,Og/a-Al2Og (0001) Fe(llI) 740 730 720 710 700 Binding Energy/ev Photo emission intensity /arb. units
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
As electrons are removed from Fe to form Fe 2 or Fe 3 ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the chemical shifts of the protons in the following compounds. (a) (b) (c) (d) (e) (f) HC(CH3)3 CH CH CH3 , - CH,CCCH OH CH,CH,-C-OH CH3 CHC-CH Br
-
The proton NMR spectrum of 2-pyridone gives the chemical shifts shown. (a) Is 2-pyridone aromatic? (b) Use resonance forms to explain your answer to (a). Also explain why the protons at (7.31and...
-
The chemical shifts of the 1H nuclei in 2,2-dimethylpropane and TMS are 0.95 and 0.0, respectively. From these data, what can you deduce about the relative electronegativities of carbon and silicon?
-
A non reactive/conservative contaminant is dumped on the ground level and it leaches to the groundwater vertically and takes half day for reaching the groundwater by travelling through unsaturated...
-
The point is on the terminal side of an angle in standard position. Find the exact values of the six trigonometric functions of the angle. 1. (12, 16) 2. (7, -24) 3. (-0.5, 4.5)
-
A toy manufacturer wants to open a third warehouse that will supply three retail outlets. The new warehouse will supply 500 units of backyard playsets per week. Two locations are being studied, N1...
-
Jackson Sales Company has prepared the following budgets for the second quarter of 2005: Required: Prepare a cash payment schedule for the second quarter of 2005. Jackson Sales Company Selling and...
-
Arantxa Corporation made the following cash purchases of securities during 2008, which is the first year in which Arantxa invested in securities. 1. On January 15, purchased 10,000 shares of Sanchez...
-
MC Qu. 107 A bond sells at a discount when the: H newconnectmheducation'com Exam 3B (40) G Help Save 5 Exit Submit 17 MC On. 107 A bond sells at a discount when the: A bond sells at a discount when...
-
You are the manager of a company that manufactures electric chainsaws. Currently the company makes 5,000 chainsaws each year and sells them for $200 each. You suspect that the company should be able...
-
The principal line in the emission spectrum of sodium is yellow. On close examination, the line is seen to be a doublet with wavelengths of 589.0 and 589.6 nm. Explain the source of this doublet.
-
Why is XPS a surface-sensitive technique?
-
Use the quadratic formula to solve each equation. (x + 1)(x-7)= 1
-
Problem 1. Cable AB is 3 m long and cable BC is 4 m long. Points A and Care at the same height. The mass of the suspended object is 359 kg. Determine the tension in cables AB and BC. .5M AR
-
Chauncey has just returned from a business trip in Britain with 150 of uncashed traveller's cheques. How much would he receive from the bank when he converts the currency back to Canadian dollars,...
-
On January 1, 2022, Pharoah Corporation acquired machinery at a cost of $1270000. Pharoah adopted the double-declining balance method of depreciation for this machinery and had been recording...
-
4) Smokey's CBD Shop has bonds outstanding with a face value of $5,000. They have 7 years left to maturity and pay annual interest of 6%. If the bonds are currently trading for a price of $4,245,...
-
Bramble Corporation bought a machine on June 1, 2019, for $29,500, f.o.b. the place of manufacture. Freight costs were $200, and $400 was spent to install it. The machine's useful life was estimated...
-
Let y i be a bin(n i =, i ) variate for group i, i = 1,..., N, with {y i } independent. Consider the model that 1 = ... = N . Denote that common value by . a. Show that the ML estimator of is p =...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
The heat capacity of chloroform (trichloromethane, CHCl 3 ) in the range 240 K to 330 K is given by C p,m /(J K 1 mol 1 ) = 91.47 + 7.5 10 2 (T/K). In a particular experiment, 1.00 mol CHCl 3 is...
-
A sample consisting of 1.0mol CaCO 3 (s) was heated to 800C, when it decomposed. The heating was carried out in a container fitted with a piston that was initially resting on the solid. Calculate the...
-
A chemical reaction takes place in a container of cross-sectional area 100 cm 2 . As a result of the reaction, a piston is pushed out through 10 cm against an external pressure of 1.0 atm. Calculate...
-
Dept of Justice, Prosecuting Computer Crimes. Chapter One Computer Fraud and Abuse Act. http://www.justice.gov/criminal/cybercrime/docs/ccmanual.pdf
-
Complete the following for this aComplete the following for this assignment Continue in the same Word file from Week 1 and under Journal 2 heading enter the date and write how you are felling right...
-
Coordinating the different stages of the value chain that become internalized within the firm results in an increase in Blank______ costs

Study smarter with the SolutionInn App


