Fill in the missing step in the derivation that led to the calculation of the spectral line
Question:
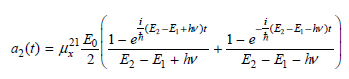
and neglecting the first term in the parentheses, show that
![оiОда, ) — Е Ги7sin'(E, - E - i)2H] (E, – E, – hv)?](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1525/3/4/0/8025aeada829f7e61525340789755.jpg)
Figure 19.24
Transcribed Image Text:
НЕ -Е -w Ez – E – lv НЕ, -Е, +i 21 Eo| 1-eй 2 а(0) %3 д 1-e E2 – E + hv оiОда, ) — Е Ги7sin'(E, - E - i)2H] (E, – E, – hv)?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
We start with Using t...View the full answer

Answered By

Ajeet Singh
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
4+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Fill in the missing particle or nucleus: (a) Ca ? + e + D (b) Cu* ? + Y gV + ? (d) 234Pu ? + a (e) Np 58 (c) Cr 46 23 24 239Pu + ? 239 93 94
-
The ketone shown was prepared in a three-step sequence from ethyl trifluoroacetate. The first step in the sequence involved treating ethyl trifluoroacetate with ammonia to give a compound A. Compound...
-
Fill in the missing parts of the following reactions. a. b. Li + on
-
1. What velocity will a 300 kg snowmobile acquire if pushed from rest by a force of 6240 N [E] for 1.25 s? What average force will stop this snowmobile from moving at this speed in 1.25 s? (v = 26.0...
-
In Exercises 1-4, determine whether the statement is true or false. Justify your answer. 1. sin(u v) = sin u cos v cos u sin v 2. cos(u v) = cos u cos v sin u sin v 3. When and are...
-
Indicate (by letter) the way each of the investments listed below most likely should be accounted for based on the information provided. Item 1. 35% of the nonvoting preferred stock of American...
-
Hal Adelman is studying for an accounting midterm examination. What should Hal know about how management may use job cost data?
-
Trinity Clinic has identified three activities for daily maternity care: occupancy and feeding, nursing, and nursing supervision. The nursing supervisor oversees 150 nurses, 25 of whom are maternity...
-
For f(x)=x and g(x)=x, find (fog)(x) and (g of)(x). Then determine whether (fog)(x) = (go f)(x). What is (fog)(x)? (fog)(x) = x What is (go f)(x)? (gof)(x) =
-
According to a 2018 article in Esquire magazine, approximately 70% of males over age 70 will develop cancerous cells in their prostate. Prostate cancer is second only to skin cancer as the most...
-
The force constants for F 2 and I 2 are 470. And 172 N m 1 , respectively. Calculate the ratio of the vibrational state populations n 1 /n 0 and n 2 /n 0 at T = 300. and at 1000.K.
-
A simulated infrared absorption spectrum of a gas-phase organic compound is shown in the following figure. Use the characteristic group frequencies listed in Section 19.5 to decide whether this...
-
What are some variations of the two-phase locking protocol? Why is strict or rigorous two-phase locking often preferred?
-
Question 2 Two spherical beads (charged equally, with each one weighing 9.50 g) are put 3.70 cm away from each other. If they are let go and begin to accelerate at 123 m/s, what would be the...
-
Cheetah Company manufactures custom-designed skins (covers) for iPods and other portable MP3 devices. Variable costs are $8.40 per custom skin, the price is $12, and fixed costs are $94,320....
-
gniwollot grid 1920A (6 Answer the following: dem od 2169y of daw lied abrod ow ysi. Two bonds both have two years to maturity, face value of $100 and pay coupons annually. The coupon rate on bond A...
-
Standish Company manufactures consumer products and provided the following information for the month of February: Units produced 131,000 Standard direct labor hours per unit 0.2 Standard variable...
-
On January 1, 2027, Talion Company purchased equipment that cost $300,000. The equipment had an expected useful life of 10 years and a residual value of $10,000. On January 1, 2031, Talion Company...
-
What is the chemical name of each of the following drugs? a. Benzocaine b. Procaine
-
QUESTION 9 HC-O-C-R R-C-O-CH HC-O-P-O-CH-CH-NH3* O || O a. Phosphatidic acid, Serine O b. Lysophosphatidic acid, Serine, Free FA O c. Lysophosphatidylserine, Free FA O d. 2 Free FAs, Serine, Glycerol...
-
Calculate the oxidation state for Cl in the compounds: a) Cl 2 O 6 b) ClF 3 c) HClO 4
-
Write the ground state electron configuration for Nb and Nb3 + .
-
How many unpaired electrons would you expect for the complex ion [Cu(NH 3 ) 6 ] 2 + ?
-
4. Qu tipo de empresas podran emplear un mercado de empresas privadas? Qu tipo de empresas podra participar en el mercado de una empresa privada? Cmo es un mercado de empresas privadas diferente de...
-
Camby Corp. can make one of the following four products: Product 1 Product 2 Product 3 Product 4 Selling price per unit $109.00 $186.00 $306.00 $45.00 Variable cost per unit $38.00 $120.00 $191.00...
-
If the order quantity (Q) for raw materials is increased, how does it affect ordering cost and/or holding cost? Group of answer choices Holding cost decreases No impact on ordering and holding costs...

Study smarter with the SolutionInn App


