For a gas that obeys the equation of state V m = Rt/P + B(T) derive the
Question:
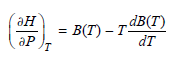
Transcribed Image Text:
dB(T) = B(T) – Tª ән т dт ӘР
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Equation 344 ...View the full answer

Answered By

Mehwish Aziz
What I have learnt in my 8 years experience of tutoring is that you really need to have a friendly relationship with your students so they can come to you with their queries without any hesitation. I am quite hardworking and I have strong work ethics. Since I had never been one of those who always top in the class and always get A* no matter what, I can understand the fear of failure and can relate with my students at so many levels. I had always been one of those who had to work really hard to get decent grades. I am forever grateful to some of the amazing teachers that I have had who made learning one, and owing to whom I was able to get some extraordinary grades and get into one of the most prestigious universities of the country. Inspired by those same teachers, I am to be like one of them - who never gives up on her students and always believe in them!
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Find an expression for the fugacity coefficient of a gas that obeys the equation of state pVm = RT(1 +B/Vm + C/V-1). Use the resulting expression to estimate the fugacity of argon at 1.00 am3 and 100...
-
Derive relations for (a) u, (b) h, and (c) s of a gas that obeys the equation of state (P + a/v 2)v = RT for an isothermal process.
-
Derive relations for (a) u. (b) h. (c) s of a gas that obeys the equation of state (P + a/v2)v = RT for an isothermal process.
-
All numbers are in $ '000. Consider an income property. Next three years its NOIs will be $25,000, $28,000 and $30,000. Then NOI will be growing at a constant rate of 3% per year. If you buy the...
-
You're leading a project to migrate customer data (biographical, billing, and orders) from the legacy system to an off-the-shelf product sold by MicroTech. The project was going well, but a key...
-
Which of the following choices best describes reasonable conclusions an analyst might make about the companys liquidity? A. Comparing FY14 with FY10, the companys liquidity improved, as indicated by...
-
Party Planners Extraordinaire Corporation has just completed operations for the year ended December 31, 2011. This is the third year of operations for the company. As the sole-shareholder, you want...
-
At the end of the current year, Accounts Receivable has a balance of $3,460,000; Allowance for Doubtful Accounts has a debit balance of $12,500; and net sales for the year total $46,300,000. Using...
-
A simple gas of mass m with a number density n and temperature T is enclosed in a container. The gas escape into a vacuum through a small circular hole of area A in the container. If the wall...
-
According to the website of the American Association for Justice,11 Stella Liebeck of Albuquerque, New Mexico, was severely burned by McDonalds coffee in February 1992. Liebeck, who re-ceived third-...
-
What is sustainable water use?
-
What can you say about H vaporization of a liquid as the temperature approaches the critical temperature?
-
Consult PCAOB Ethics and Independence Rule 3520. What is auditor independence, and what is its significance to the audit profession? What is the difference between independence in appearance and...
-
A. What are some characteristics of the Social Age? B. What are some strategies for organizing a dashboard to help you work more efficiently with your teams? C. What do you think are some of the most...
-
Two identical particles of inertia \(m\) collide elastically on a low-friction table. Calculate the kinetic energy of the system before and after the collision in the zero-momentum frames.
-
Revise the entire student club flyer. Create an effective flyer to help promote the Supply Chain Management Club. Make sure the document is as easy to read as possible while retaining all relevant...
-
A horizontal escalator connecting airport terminals is \(1 \mathrm{~km}\) long. Two kids start from opposite ends and run toward each other at \(3 \mathrm{~m} / \mathrm{s}\) and meet \(600...
-
Does a man standing in an elevator that is moving upward at a constant speed of \(10 \mathrm{~m} / \mathrm{s}\) feel heavier or lighter? What if the elevator is moving downward at the same speed? If...
-
The government net financial liabilities as a percentage of GDP is given in the table below for selected countries in 2015 according to stats.oecd.org. Present these data in a graph that allows you...
-
Identify the source of funds within Micro Credit? How does this differ from traditional sources of financing? What internal and external governance mechanisms are in place in Micro Credit?
-
Calculate the molar volume of chlorine gas at 350 K and 2.30 atm using (a) The perfect gas law and (b) The van der Waals equation. Use the answer to (a) to calculate a first approximation to the...
-
Calculate the volume occupied by 1.00 mol N2 using the van der Waals equation in the form of a virial expansion at (a) Its critical temperature, (b) Its Boyle temperature, and (c) Its inversion...
-
The mass density of water vapour at 327.6 atm and 776.4 K is 133.2 kg m-3. Given that for water Tc = 647.4 K, Pc = 218.3 atm, a = 5.464 dm6 atm mol-2, b= 0.03049 dm3 mol-1, and M= 18.02 g mol-1,...
-
Ridge Crest Company has beginning Retained Earnings of $23,000, ending Retained Earnings of $32,600, and net income of $15,500. What was the amount of dividends declared during the year?
-
Solomon Manufacturing Company established the following standard price and cost data. Sales price Variable manufacturing cost Fixed manufacturing cost $ 8.50 per unit Fixed selling and administrative...
-
Shadee Corporation expects to sell 640 sun shades in May and 380 in June. Each shade sells for $134. Shadee's beginning and ending finished goods inventories for May are 70 and 40 shades,...

Study smarter with the SolutionInn App


