From the following data at 298.15 K as well as data in Table 4.1 (Appendix B, Data
Question:
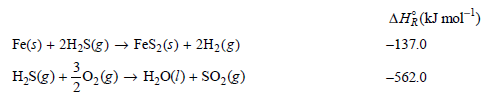
Transcribed Image Text:
AĦR(kJ mol) Fe(s) + 2H2S(g) –→ FeS2(s) + 2H2(g) -137.0 H;S(g) + 0,(g) → H,O() + SO,(E) -562.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
HOl SOg HSg 20 8 Ss Og SO g Hg Og HO1 ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the standard enthalpy of formation for diamond, given that C(graphite) + O2(g) CO2(g) Afr =-393.5 kJ/mol C(diamond) + O2(g) CO2(g) AF1 395.4 kJ/mol
-
From the following data at 298.15 K calculate the standard enthalpy of formation of FeO(s) and of Fe 2 O 3 (s): A(kJ mol) Fe,0;(s) + 3C(graphite) 2Fe(s) + 3cO(g) FeO(s) + C(graphite) Fe(s) + CO(g)...
-
From the enthalpy of formation for CO2 and the following information, calculate the standard enthalpy of formation for carbon monoxide (CO). Why can't we obtain it directly by measuring the enthalpy...
-
The following items are dropped from an airplane. Rank them in order from lowest terminal speed to highest and justify your ranking. (a) Bowling ball (b) Beach ball (c) Spear or javelin (pointing...
-
San Antonio Company produces accounting software. Its unit cost structure, based on an anticipated production volume of 150,000 units, is:sale price is 160, variable costs is 60 and fixed costs is...
-
In the following take CV = 20.8 and CP = 29.1 J mol-1 oC-1 for nitrogen gas: (a) Three moles of nitrogen at 30oC contained in a rigid vessel, is heated to 250oC. How much heal is required if the...
-
Why is the sales forecast the starting point for budgeting?
-
Ironjay, Inc., produces two types of weight-training equipment: the jay-flex (a weight machine that allows the user to perform a number of different exercises) and a set of free weights. Ironjay...
-
Question 20 0 out of 3.03 points John won a lottery that will pay him $150,000 at the end of each of the next twenty years. Assuming an appropriate interest rate is 8% compounded annually, what is...
-
1. How can Diva demystify what happens behind the scenes and make potential Generation Y employees aware of the opportunities available to them beyond the shop floor? 2. Diva has implemented a...
-
Which of Ne or Ar has the larger van der Waals parameter a? Explain your reasoning.
-
Which of Ne or Ar has the larger van der Waals parameter b? Explain your reasoning.
-
What is the port number for SMTP proto- col? a. a. 25 b. b. 23 c. c. 70 d. d. 80
-
Design Galore currently sells polo shirts for $1,875 which includes a margin of 20%. Each order is placed by a single individual that charges an average rate of $405 per hour. To file the paper works...
-
Compute the following current (2019-2020) financial ratios for both companies (Home Depot and Lowes): Liquidity measurement ratio: Current ratio Profitability indicator ratios:Return on assetsReturn...
-
9. Jenny received two endorsement offers Under Armour offers $50,000 to be paid, in a single payment, in 5 years and Nike negotiated a long-term deal that called for yearly payments to Jenny of...
-
Suppose AMAZON issued a 20-year 10% coupon bond eight years ago. Today, the bond is selling at 80% of its face value, that is, at $ 800. What is the cost of AMAZON debt? (Use 4 decimal places, do not...
-
Consider an expression of the form . ax2 + bx + c 0 (mod N) 1. Show that the 's of the following form are solutions of the above system:. x (b b2 4ac) (2a) (mod N) when and is a Quadratic Residue mod...
-
Audrey also has a sample of fraternal twin pairs. She gives them the same test of extraversion to the fraternal twins and finds that D = 1.34, s D = 2.40, and n D = 15. a. What is the null...
-
Differentiate the following terms/concepts: a. Personality types and money attitudes b. Planners and avoiders c. Moderating and adapting to biases d. "Perfectible judges" and "incorrigible judges"
-
A lot of information about the energy levels and wave functions of small inorganic molecules can be obtained from their ultraviolet spectra. An example of a spectrum with considerable vibrational...
-
Aromatic hydrocarbons and 12 form complexes from which charge transfer electronic transitions are observed the hydrocarbon acts as an electron donor and 12 as an electron acceptor the energies hvmax...
-
Consider some of the precautions that must be taken when conducting single-molecule spectroscopy experiments. (a) What is the molar concentration of a solution in which there is, on average, one...
-
hello , can you please help me answer those a question correctly, thank you. Saved Help Save & Exit Subn Keisha owns 100% of DEF Company and has a basis ofof $12,000. Keisha has a: $10,000 in its...
-
Accountants that are focused on issuing financial statements to serve the dicision making needs of external users of the business are called
-
Under the default provisions, the financial rights between member-management owners and manager-management owners is: Question 2 options: a) immaterial. b) material, giving a higher percentage of...

Study smarter with the SolutionInn App


