Identify the molecular orbitals for F 2 in the images shown here in terms of the two
Question:
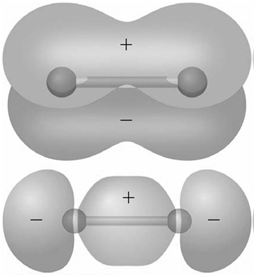
Transcribed Image Text:
+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (10 reviews)
The top image is of symmetry and has no ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Draw the molecular orbitals for the cyclopropenyl case. (Since there are three p orbitals, there must be three MOs: one all-bonding MO and one degenerate pair of MOs.) (b) Draw an energy diagram...
-
Evaluate the Klopp case in terms of those two criteria.
-
Images of molecular orbitals for LiH calculated using the minimal basis set are shown here. In these images, the smaller atom is H. The H1s AO has a lower energy than the Li2s AO. The energy of the...
-
9:49 X Bank Reconciliation Assignment 1 PDF 150 KB Assignment #1 - Bank Reconciliation This assignment has 33 total marks. This assignment is worth 5% of your final grade. This assignment is due...
-
A police car with its spotlight on is parked 20 meters from a warehouse. Consider and x as shown in the figure. (a) Write θ as a function of x. (b) Find θ when x = 5 meters...
-
Zhou Inc. operates a retail operation that purchases and sells snowmobiles, among other outdoor products. The company purchases all inventory on credit and uses a periodic inventory system. The...
-
Discuss why only relevant costs need to be considered in a short-term decision.
-
Where would each of the following items most likely be reported in a companys financial statements? Assume the monetary amount of each item is material. 1. Bad debts expense 2. Sales discounts taken...
-
Suppose we want to evaluate the definite integral, r2502 - 4r2 dr using the substitution, = Part 1. 16 2502-4r2 Re-write the definite integral in terms of the variable u and remember to use the...
-
Wong Inc., the lessee entered into two leases on July 1, 2020 with Pomerleau Corp. Both companies are public corporations following IFRS. The leases are for a large auger and a jackhammer that will...
-
If there is a node in u , is the electron in this wave function really delocalized? How does it get from one side of the node to the other?
-
By considering each term in And Explain why the values of J and K are positive for H + 2 . e? |e, dr - j (, Hs, dt J = [@n| dt 4TEQ7B.
-
How do reserve requirements on bank deposits affect a bank's ability to create money? What would happen to the money supply if a 100 percent reserve requirement were established?
-
A technology company needs to send a specific number of mobile phones to its sales center to meet the demand for the next three months. The required quantities to be sent are 300 units for the first...
-
Nelson Manufacturing uses a process costing system with 2 departments, Assembly and Packaging. Units in process in the Assembly Department at the beginning of the period totaled 2 , 0 0 0 . During...
-
Carmen's Beauty Salon has estimated monthly financing requirements for the next six months as follows: January February March $ 9,000 3,000 4,000 April May June $ 9,000 10,000 5,000 Short-term...
-
Evaluate the value that technology offers organizations and to customers. Are technologies enablers of innovation? Next, analyze innovation. How is innovation developed in response to customer needs?...
-
A child's ball (a thin hollow uniform sphere) of mass 34.8 g and radius 0.141 m starts from rest and rolls without slipping down an inclined plane of vertical height 6.82 m. What is the angular speed...
-
What monosaccharides form the same osazone as D-sorbose?
-
a. What is meant by the term tax haven? b. What are the desired characteristics for a country if it expects to be used as a tax haven? c. What are the advantages leading an MNE to use a tax haven...
-
The CH 3 Cl molecule belongs to the point group C 3v . List the symmetry elements of the group and locate them in the molecule.
-
Show that, for a perfect gas, (U/S) V = T and (U/V) S = p.
-
Sketch the form of the 19 F-NMR spectra of a natural sample of tetrafluoroborate ions, BF 4 , allowing for the relative abundances of 10 BF 4 and 11 BF 4 .
-
The consolidated financial statements can be prepared by using which procedures? Question 1Select one: a. Pro forma consolidation journal entries prepared from the analysis of owners' equity and then...
-
Harrison Rentals Company faced the following situations. (Click the icon to view the situations.) Requirement 1. Journalize the adjusting entry needed at December 31, 2023, for each situation....
-
Emma holds a $5,000 portfolio that consists of four stocks. Her investment in each stock, as well as each stock's beta, is listed in the following table: Stock Investment Beta Standard Deviation Omni...

Study smarter with the SolutionInn App


