Is the expression only valid for an ideal gas if V is constant? T, AUr = [
Question:
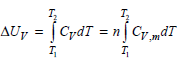
only valid for an ideal gas if V is constant?
Transcribed Image Text:
T, AUr = [ CydT = n[ Cy,maT
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
No It is always valid for an ...View the full answer

Answered By

Muhammad Mahtab
everyone looks that their work be perfect. I have more than a five year experience as a lecture in reputable institution, national and international. I provide perfect solution in marketing, case study, finance problems, blog writing, article writing, business plans, strategic management, human resource, operation management, power point presentation and lot of clients need. Here is right mentor who help clients in their multi-disciplinary needs.
5.00+
3+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Why is the equation
-
Is the equation valid for an ideal gas? Tf PV; -V;) T; Cy dT Lav = C, n2v, -v) %3D AS =
-
Show that for an ideal gas cp = cv + R u.
-
Each of the following passages may be plausibly criticized by some who conclude that it contains a fallacy, but each may be defended by some who deny that the argument is fallacious. Discuss the...
-
When reviewing a balance sheet of a corporation, what are things or items that indicate a company is in good financial health? What items indicate poor financial health? How can these concepts be...
-
f (z) is the branch z1+i = exp[(1 + i)log z] (|z| > 0, 0 < argz < 2) of the indicated power function, and C is the unit circle z = ei (0 2).
-
What is a concentration account?
-
Eon Industries is deciding whether to automate one phase of its production process. The manufacturing equipment has a six-year life and will cost $920,000. Projected net cash inflows are as follows:...
-
Based on the following data, would Ann and Carl Wilton receive a refund or owe additional taxes? (Input the amount as a positive value. Round your intermediate calculations and final answer to the...
-
Mr B aged 52 years, has earned rupees 75,00,000 out of his business. His ex-wife gifted him a car worth rupees 8 lakh. He spent a total of rupees 20 lakh during a family trip. He won a lottery of 16...
-
For each pair of compounds below, identify the one that would be expected to have more ionic character. Explain your choice. a) NaBr or HBr b) BrCl or FCl
-
Draw a Lewis dot structure for each of the following compounds: a. CH 3 CH 2 OH b. CH 3 CN
-
Is it desirable to eliminate natural unemployment? Why or why not? Hint: Think about what our economy would look like today and what assumptions would have to be met to have a zero rate of natural...
-
a. Construct the Bode plot for the transfer function b. Draw the Bode plots for H(w) = 200jw (jw+2)(jw+ 10)
-
This is a capstone forum requiringconsolidation of information learned and skills acquired during the courseterm. Here you will share with classmates a research study proposal you havedeveloped that...
-
The advanced practice psychiatric nurse may intervene as appropriate with agencies or persons in order to advocate for the patient; do for the patient what he or she cannot do for self, always with...
-
5. Demarius has just moved, and is unloading boxes the moving van. He slides a 19.0 kg box down the 31 ramp. The box accelerates at a rate of 0.68 m/s. What is the coefficient of kinetic friction...
-
Briefly describe the problem, the hypothesis, procedure and results of the study. 2. Do you see any potential problems with this study, ethical concerns, methodical issues? 3. Do you agree with the...
-
Suppose that there is a collection of 3n distinct coupons, n of which are colored red and 2n of which are colored blue. Suppose that each time you go to a ticket window to get a coupon, the clerk...
-
Suppose that the laptop of Prob. 2.16 is placed in an insulating briefcase with a fully charged battery, but it does not go into sleep mode, and the battery discharges as if the laptop were in use....
-
The effective rate constant for a gaseous reaction that has a Lindemann-Hinshelwood mechanism is 1.7 X 10-3 s-I at 1.09 kPa and 2.2 X 10-4 S-1 at 25 Pa. Calculate the rate constant for the activation...
-
The data below apply to the formation of urea from ammonium cyanate, NH4CNO --7 NH2CONH2. Initially 22.9 g of ammonium cyanate was dissolved in enough water to prepare 1.00 dm3 of solution. Determine...
-
The thermal decomposition of an organic nitrile produced the following data: Determine the order of the reaction and the rate constant. t/(10's) 0 2.00 4.00 6.00 8.00 10.00 12.00 [nitrile]/(mol dm)...
-
The transactions listed below are typical of those involving New Books Incorporated and Readers Corner. New Books is a wholesale merchandiser and Readers Corner is a retail merchandiser. Assume all...
-
Oriole, Inc. uses a flexible budget for manufacturing overhead based on machine hours. Variable manufacturing overhead costs per machine hour are as follows: Indirect labor Indirect materials...
-
Mac Users This exam assesses your Access technical knowledge Access Chapters 1-3 PowerPoint provided representations of Access screens Using this, plus the class recordings and other resources as the...

Study smarter with the SolutionInn App


