Predict which of the bent molecules, BH 2 or NH 2 , should have the larger bond
Question:
Figure 24.11
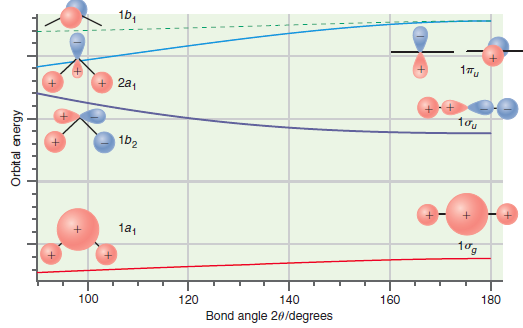
Transcribed Image Text:
16, 1Tu + 2a, 1b2 1a1 180 160 140 100 120 Bond angle 20/degrees Orbital energy
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
Both molecules are equivalent through the 2a 1 1 orbital Howev...View the full answer

Answered By

Aijaz Khan
I am highly enthusiastic about tutoring. I share a friendly but professional relationship with my students. After completing my electrical engineering I actually taught a course to undergraduates for GATE exam as T.A and it was a brilliant experience and I was one among very few to finish my course on time. I have also helped professors to prepare lessons for my junior fellows while doing my undergrad. Every time I have taught so far, the response has been very heart warming. I hope to continue this and keep on improving it till I am here. Apart from this I have conducted many one on one tutoring lessons.
I believe in focussing on basic and core concepts inorder to keep students' interest alive. My only aim while tutoring is to make student understand concept in such a way that he/she can explain the learnt topic to anyone. Apart from this problem solving is my main focus while tutoring.
I hope to work with SolutionInn for long time. Hope my students will feel the difference.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict whether the ground state or the first excited state of CH 2 should have the larger bond angle on the basis of the Walsh correlation diagram shown in Figure 24.11. Explain your answer. 1b, 17u...
-
Suppose we have some optically pure (R)-2-butyl acetate that has been "labeled" with the heavy 18O isotope at one oxygen atom as shown. (a) Draw a mechanism for the hydrolysis of this compound under...
-
Predict which of the following liquids has greater surface tension: ethanol (C2H5OH) or dimethyl ether (CH3OCH3)?
-
How well employees modify their thoughts and behavior to align with and support a new or changing environment is known as Multiple Choice proactive task performance. proficient task performance....
-
Find the exact values of the remaining trigonometric functions of satisfying the given conditions. 1. tan = 15/8, sin > 0 2. cos = 8/17, tan < 0
-
Mississippi Products Co. had the following positive cash flows during the current year: received cash from customers of $750,000; received bank loans of $35,000; and received cash from the sale of...
-
The following information for Great Oaks Furniture, a retail furniture and design firm, relates to Exercises 6 through 11. Calculate asset turnover, accounts receivable turnover, days sales in...
-
Manno Corporation has the following information available concerning its postretirement benefit plan for 2010. Service cost $40,000 Interest cost 47,400 Actual and expected return on plan assets...
-
Hi could you please help with the following question. You wish to sell a 240 day Note that pays $68,000 at maturity. Simple interest rate is 8.32% pa. If the sales occurs 52 days before maturity,...
-
A manufacturing company produces steel housings for electrical equipment. The main component part of the housing is a steel trough that is made out of a 14- gauge steel coil. It is produced using a...
-
Show that the water hybrid bonding orbitals given by a = 0.55 2Pz + 0.71 2 px -0.45 2 ps b = 0.55 2pz - 0.71 2px - 0.45 2s are orthogonal.
-
Derive two additional mutually orthogonal hybrid orbitals for the lone pairs on oxygen in H 2 O, each of which is orthogonal to Ï a and Ï b , by following these steps: a. Starting with the...
-
Explain how the thyroid and parathyroid glands work together to maintain blood calcium homeostasis?
-
Evaluate the definite integral 5 L (8x+6)dz skmistor
-
Analyze the challenges and techniques involved in process migration across distributed systems. How do operating systems and distributed computing frameworks manage process state, data consistency,...
-
Determine (a) the $ net markdown and (b) the net markdown % from the following figures: Gross Sales $450,000 Customer Returns $10,000 Total Markdowns $124,800 Markdown Cancellation. $32,400
-
Oriole Railroad Co. is about to issue $260,000 of 10-year bonds paying an 10% interest rate, with interest payable annually. The discount rate for such securities is 8%. Click here to view the factor...
-
Explore the security mechanisms in place to isolate processes and prevent unauthorized access to system resources. How do modern operating systems use features like chroot, namespaces, and privilege...
-
Snow Cap Springs produces and sells water filtration systems for homeowners. Information regarding its three models is shown below. The company's total fixed costs to produce the filtration systems...
-
The following table shows the rates of total return in successive years from 2004 to 2008 for the Sprott Canadian Equity Fund and for the benchmark Toronto Stock Exchange S&P/TSX Composite Index. By...
-
Derive an expression for the time dependence of the degree of polymerization for a stepwise polymerization in which the reaction is acid catalysed by the -COOH acid functional group. The rate law is...
-
Calculate the average polymer length in a polymer produced by a chain mechanism in which termination occurs by a disproportionation reaction of the form M + M M + :M.
-
Calculate the ratio of the mean cube molar mass to the mean square molar mass in terms of (a) The fraction p, (b) The chain length.
-
Eleanor is opening a store called "Everything is $10". As the name implies, all of the items sold at their shop will be priced at $10. They are estimating their variable cost per item they sells will...
-
Match the letter with the property of the improper integral. A The integral converges. B The integral diverges. Submit Answer Tries 0/8 3 L 6 dx x L 6 8 dx (x-3)4/5 3. S La Lo S 6 dx (3-x)4 6 34 dr...
-
Match the basic step in the recording process described by entering the appropriate statement number in the space provided. A. Analyze each transaction B. Enter each transaction in a journal C....

Study smarter with the SolutionInn App


