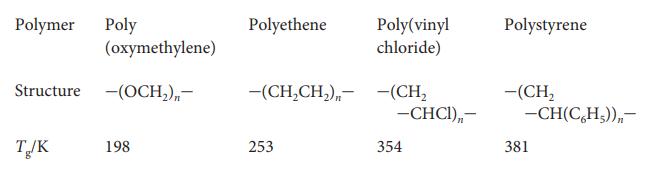
The following table lists the glass transition temperatures, T g , of several polymers. Discuss the reasons
Question:
The following table lists the glass transition temperatures, Tg, of several polymers. Discuss the reasons why the structure of the monomer unit has an effect on the value of Tg.
Transcribed Image Text:
Polymer Poly (oxymethylene) Structure -(OCH₂),- T/K 198 Polyethene Poly(vinyl chloride) -(CH₂CH₂)--(CH₂ 253 -CHCI),,- 354 Polystyrene -(CH₂ -CH(C₂H₂)),- 381
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
The structure of the monomer unit has a significant effect on the glass transition temperature Tg of ...View the full answer

Answered By

Dansteve Matoke
As a consequence of more than three years of experience in ACADEMIC WRITING, I have vast, diverse knowledge and impeccable grammar. I guarantee the quality of my work across multiple fields. I intend to precisely meet the clients' expectations. Let's work together to achieve exceptional grades.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Physical Chemistry Thermodynamics And Kinetics
ISBN: 9781464124518
10th Edition
Authors: Peter Atkins, Julio De Paula
Question Posted:
Students also viewed these Sciences questions
-
The following table lists several characteristics. Place a check mark next to those items which pertain directly to the internal audit function and its role within the organization. Characteristic...
-
The following table lists several characteristics. Place a check mark next to those items that pertain directly to the internal audit function and its role within the organization. Characteristic...
-
The following table lists the earnings per event that were referred to in Exercise 2.89. Although the table lists earnings per event, players are listed in order of their total earnings, not their...
-
Consider a random walk consisting of equi-probable p = q = 1/2 steps in left or right directions. However the step length at ith step is given by e-, i = 1,2,3..... N, with > 0 a constant. Calculate...
-
There are companies that have traditionally been known for their excellence in tangibles, e.g., Xerox in copiers, IBM in computers, who now describe themselves as primarily service companies. Do you...
-
Consider four independent events A1, A2, A3, and A4, and let pi = P(Ai) for i = 1,2,3,4. Express the probability that at least one of these four events occurs in terms of the pis, and do the same for...
-
Summarize ways you can lower lender risk to increase your chances of getting a loan at a lower interest rate?
-
Morse Company manufactures basketballs. Materials are added at the beginning of the production process and conversion costs are incurred uniformly. Production and cost data for the month of July 2012...
-
Using the perpetual inventory system, prepare general journal entries for the following selected transactions of Silver Traders in July. Ignore GST. 5 July Purchased inventory from Crane Ltd in cas...
-
Holz Disc Golf Course was opened on March 1 by Ian Holz. The following selected events and transactions occurred during March. Mar. 1 Invested $20,000 cash in the business. 3 Purchased Rainbow Golf...
-
Distinguish between elastic and plastic deformation.
-
Develop an expression for the fundamental vibrational frequency of a one-dimensional random coil that has been slightly stretched and then released. Evaluate this frequency for a sample of polyethene...
-
During the most recent month, Fabricators Inc. started 3,000 units, finished 3,100 units, and incurred conversion costs totaling $290,160. The finished goods ending inventory consisted of 50 units....
-
A. You are offered to buy a 4-year coupon corporate bond in the beginning of its 7th month on its third year for $963.94. Its face value is $1,000 and its coupon rate is 5.172% p.a. with coupon paid...
-
Explain the differences between a traditional and activity based costing system for use in a service organisation.
-
West End Industrial Gas Corporation supplies acetylene and other compressed gases to industry. Data regarding the store's operations follow: Sales are budgeted at $370,000 for November, $390,000 for...
-
You are constructing a portfolio of two assets, Asset A and Asset B . The expected returns of the assets are 1 2 percent and 1 6 percent, respectively. The standard deviations of the assets are 3 6...
-
The following information relates to production activities of Mercer Manufacturing for the year. Accual direct materials used Actual direct labor used Actual units produced Standard quantity and...
-
On January 1, 2016, the controller of Omicron Inc. is planning capital expenditures for the years 2016-2019. The following interviews helped the controller collect the necessary information for the...
-
The following table shows the rates of total return in successive years from 2004 to 2008 for the Sprott Canadian Equity Fund and for the benchmark Toronto Stock Exchange S&P/TSX Composite Index. By...
-
Predict the number of chemically shifted 1 H peaks and the multiplet splitting of each peak that you would observe for 1,1,1,2-tetrachloroethane. Justify your answer. I H1 --c-CI CI | Cl
-
Predict the number of chemically shifted 1 H peaks and the multiplet splitting of each peak that you would observe for 1, 1, 2, 2-tetrachloroethane assuming that there is no rotation of the two...
-
Predict the number of chemically shifted 1 H peaks and the multiplet splitting of each peak that you would observe for nitroethane. Justify your answer.
-
Which one of the following statements accurately describes the results or duration of the nonimportation movement? a . It resulted in an economic recession in England that led many British citizens...
-
Question 6 ________ management involves purchasing, engineering, supplier quality assurance, the supplier, and other related functions working together to further mutual goals. A System B Supply C...
-
Question 11 A feasibility study should consider which three elements? Design, operations, and location O Marketing, location, and finances O Facility, location, and budget O Location, location,...

Study smarter with the SolutionInn App


