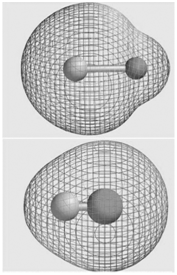
The molecular electrostatic potential maps for LiH and HF are shown here. Does the apparent size of
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Yes it does The electrostatic potential is displayed on a tot...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the molecular electrostatic potential map for the NH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the BH 3 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
Consider the molecular electrostatic potential map for the BeH 2 molecule shown here. Is the hydrogen atom (shown as a white sphere) an electron acceptor or an electron donor in this molecule?
-
What was the action you are most proud of, or that is the most meaningful to you? What was the civic issue that was involved in your action? When and how did you first hear about that issue? Why did...
-
In Exercises 1-4, find the exact value of the expression, if possible. 1. sin(arctan 3/4) 2. cos(arcsin 4/5) 3. cos(tan 12) 4. sin(cos 15)
-
Indicate in which financial statement each of the following adjusted trial balance yet account would be presented. Service Revenue ....................... Accounts Receivable Notes Payable...
-
On a specific flight there are 240 seats available, at a maximum price of 200 per seat. However only 180 seats are sold, with an average achieved ticket price of 110. Calculate the yield management %...
-
WJTY Company issued $300,000, 11%, 10-year bonds on January 1, 2012, for $318,694. This price resulted in an effective-interest rate of 10% on the bonds. Interest is payable semiannually on July 1...
-
An individual has $100000 in income now, but no income in the future. The current interest rate is 5%. We observe that the individual consumes $50000 now and will consume $52500 in the future. Assume...
-
You know that if a tax is imposed on a particular product, the burden of the tax is shared by producers and consumers. You also know that the demand for automobiles is characterized by a stock...
-
Why is it reasonable to approximate H 11 and H 22 by the appropriate ionization energy of the corresponding neutral atom?
-
For H + 2 , explain why H aa is the total energy of an undisturbed hydrogen atom separated from a bare proton by the distance R.
-
Distinguish between centralized and decentralized P2P networks.
-
Let X if x < 1 3 if x = 1 g(x) = 2 x2 if 1 < x 2 x - 3 if x > 2 Evaluate each of the following, if it exists. a) lim g(x) x1- b) lim g(x) x-1 c) g(1) d) lim g(x) x+2 e) lim g(x) x+2+ f) lim g(x) X2
-
Pool Accessories, Inc., has two divisionsFurniture and Supplies. Assume for both divisions that the tax rate is 30 percent, and the cost of capital is 8%. The following segmented financial...
-
Let f(x) = 5. Simplify each of the following expressions. f(4) 54 f(4+ h) = f(4 + h) f(4) = f(4 + h) f(4) h = (Make sure to combine terms by getting a common denominator.) (Make sure to cancel out...
-
Automated material handling systems, manufacturing assembly lines and autonomous industrial robotics are typically designed to perform superposition motion in a vertical plane. The path of motion is...
-
Equal employment opportunity and subsequent discrimination are major factors in today's workplace. For a quick review, you can access the U.S. Equal Employment Opportunity Commission's Discrimination...
-
The following transactions have been encountered in practice. Assume that all amounts are material. a. A company decided to put the assets of one product line up for sale (intended to be sold within...
-
Walker, Inc., is an all-equity firm. The cost of the company's equity is currently 11.4 percent and the risk-free.rate is 3.3 percent. The company is currently considering a project that will cost...
-
The molar absorption coefficient of a substance dissolved in hexane is known to be 855 dm 3 mol 1 cm 1 at 270 nm. Calculate the percentage reduction in intensity when light of that wavelength passes...
-
The term symbol for the ground state of N 2 + is 2 g + . What is the total spin and total orbital angular momentum of the molecule? Show that the term symbol agrees with the electron configuration...
-
Explain the origin of the term symbol 3 g for the ground state of dioxygen.
-
Rosie Dry Cleaning was started on January 1, Year 1. It experienced the following events during its first two years of operation: Events Affecting Year 1 Provided $34,850 of cleaning services on...
-
The IMA's Statement of Ethical Professional Practice includes Standards of conduct for Management Accountants. The standard that includes management accountants responsibility to provide all relevant...
-
At the beginning of the period, the Cutting Department budgeted direct labor of $63,920 and supervisor salaries of $47,190 for 3,760 hours of production. The department actually completed 4,100 hours...

Study smarter with the SolutionInn App


