Use the Davies equation to calculate for a 1.00 molar solution of KOH. Compare your
Question:
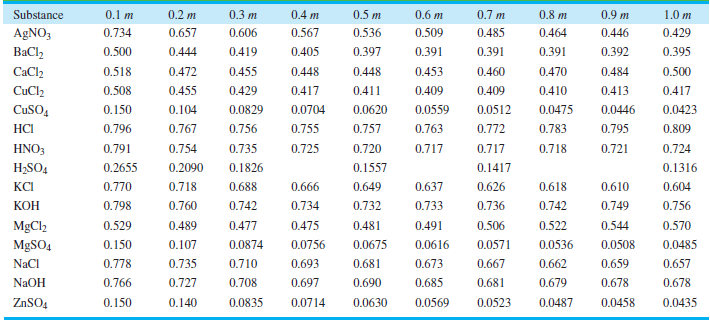
Use the Davies equation to calculate γ±for a 1.00 molar solution of KOH. Compare your answer with the values in Table 10.3.
Transcribed Image Text:
0.2 m 0.7 m 0.4 m 0,8 m 0.9 m Substance 0.1 m 0.3 m 0.5 m 0.6 m 1.0 m 0.657 0.485 AGNO3 0.734 0.606 0.567 0.536 0.509 0.464 0.446 0.429 0.405 0.391 BaCl, 0.500 0.444 0.419 0.397 0.391 0.391 0.392 0.395 CaCl2 0.518 0.472 0.455 0.448 0.448 0.453 0.460 0.470 0.484 0.500 0.508 0.411 0.409 0.413 CuCl2 0.455 0.429 0.417 0.409 0.410 0.417 0.0423 CuSO, 0.150 0.104 0.0829 0.0704 0.0620 0.0559 0.0512 0.0475 0.0446 0.755 0.763 0.783 0.795 НC 0.796 0.767 0.756 0.757 0.772 0.809 0.791 0.725 HNO3 0.754 0.735 0.720 0.717 0.717 0.718 0.721 0.724 H2SO4 0.2655 0.2090 0.1826 0.1557 0.1417 0.1316 КСі 0.770 0.718 0.688 0.666 0.649 0.637 0.626 0.618 0.610 0.604 0.798 КОН 0.760 0.742 0.734 0.732 0.733 0.736 0.742 0.749 0.756 0.475 MgCl2 0.529 0.489 0.477 0.481 0.491 0.506 0.522 0.544 0.570 0.0756 0.0616 MgSO4 0.150 0.107 0.0874 0.0675 0.0571 0.0536 0.0508 0.0485 0.693 0.673 NaCl 0.778 0.735 0.710 0.681 0.667 0.662 0.659 0.657 NAOH 0.766 0.697 0.727 0.708 0.690 0.685 0.681 0.679 0.678 0.678 0.140 0.0630 0.0569 0.0435 ZNSO4 0.150 0.0835 0.0714 0.0523 0.0487 0.0458
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
This answer is reasonably c...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the pH of a buffer solution that is 0.200 molal in CH 3 COOH and 0.15 molal in CH 3 COONa using the Davies equation to calculate . What pH value would you have calculated if you had...
-
From pK1 and pK2 for glycine at = 0 in Table 9-1, compute pK1 and pK2 that apply at = 0.1 M. Use the Davies equation for activity coefficients. Compare your answer with experimental values in cells...
-
The solution containing no added KNO3 for Figure 7-1 contains 5.0 mM Fe(NO3)3, 5.0 M NaSCN, and 15 mM HNO3. We will use Davies activity coefficients to find the concentrations of all species in the...
-
Split the number 750 into two numbers x and y so that the sum of 8% of the first number and 24% of the second number is 11.2% of the sum of x and y.
-
Ingram Co. manufactures office furniture. During the most productive month of the year, 3,552 desks were manufactured at a total cost of $83,771. In its slowest month, the company made 1,243 desks at...
-
A rectangle is to be inscribed in a semicircle of radius r, as shown in Figure 25. What are the dimensions of the rectangle if its area is. to be maximized?
-
Boswell Enterprises Ltd is reviewing its trade credit policy. The business, which sells all of its goods on credit, has estimated that sales revenue for the forthcoming year will be 3 million under...
-
A container of car antifreeze is supposed to hold 3785mL of the liquid. Realizing that fluctuations are inevitable, the quality-control manager of the Taconic Chemical Company wants to be quite sure...
-
Suppose we wish to train a classification tree to predict whether a new smartphone will be profitable on Amazon.com. To build the classification tree, we have a dataset which consists of hundreds of...
-
As a group you are the senior teaching staff of a school where each subject department is regarded as a cost centre. The direct costs of each cost centre are teachers salaries, books and worksheets...
-
What are the major differences between the positivist and interpretivist paradigms in consumer research?
-
Each country or region will have a core of products and services that are consumed. Collectively, the consumption of these infers some kind of group bond. Identify examples of these specific products...
-
Evaluate the integral along the path C. C: line segments from (0, 0) to (0, -3) and (0, -3) to (2, -3) (((2x - y) (2x - y) dx + (x + 3y) dy C
-
Describe, in your own words, the various uses of QuickBooks Accountant.
-
Describe the function of the centers in QuickBooks Accountant.
-
Discuss the importance of the sales budget in the corporate budgetary process.
-
Name three forms used in QuickBooks Accountant.
-
Ryan's Wedding Services has been open for one year, and Shu Ryan, the owner, wants to know whether the business earned a net income or a net loss for the year. First, she must identify the revenues...
-
A small water droplet in a mist of air is approximated as being a sphere of diameter 1.5 mil. Calculate the terminal velocity as it falls through still air to the ground. Is it reasonable to neglect...
-
Avatar Financials, Inc., located on Madison Avenue, New York City, is a company that provides financial advice to individuals and small- to mid-sized businesses. Its primary operations are in wealth...
-
Label the regions of the phase diagram in Fig. 6.38. State what substances (if compounds give their formulas) exist in each region. Label each substance in each region as solid, liquid, or gas. Fig....
-
Haemoglobin, the red blood protein responsible for oxygen transport, binds about 1.34 cm 3 of oxygen per gram. Normal blood has a haemoglobin concentration of 150 g dm 3 . Haemoglobin in the lungs is...
-
K. Sato, F.R. Eirich, and J.E. Mark (J. Polymer Sci., Polym. Phys. 14, 619 (1976)) have reported the data in the table below for the osmotic pressures of polychloroprene ( = 1.25 g cm 3 ) in toluene...
-
Pregunta 51 pts Aldo es un joven profesor del rea de Matemticas que, actualmente, trabaja en un colegio de la capital de Honduras. En una de sus clases, comparti con sus estudiantes que el valor del...
-
Bristol Syrup Company Ltd (BSC) makes a unique syrup using cane sugar and local herbs. The syrup is sold in small bottles and is prized as a flavouring for drinks and for use in desserts. The bottles...
-
Casey Nelson is a divisional manager for Pigeon Company. His annual pay raises are largely determined by his divisions return on investment (ROI), which has been above 23% each of the last three...

Study smarter with the SolutionInn App


