Using your results from Problem P5.7, calculate q, ÎU, and ÎH for each step in the cycle
Question:
Figure 5.2
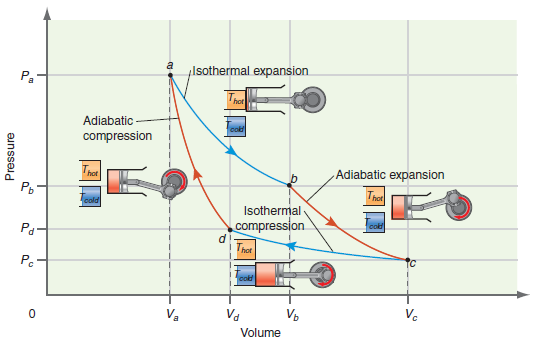
Transcribed Image Text:
Isothermal expansion Pa Adiabatic cold compression Adiabatic expansion hot Isothermal compression Pg- hot Pc Va Va Vo Vc Volume Pressure
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
ab U H 0 because T 0 q w 944 10 3 J bc U w 112 10 4 J be...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Using your results from Problem P24.10, a. Calculate the s and p character of the water lone pair hybrid orbitals b. Show that the lone pair orbitals are orthogonal to each other and to the hybrid...
-
Using your results from Problem 3.8, find the heat flow at the base of the fin for the following conditions:k = 20 Btu/(h ft ?F)L = 2 in. ho = 20 Btu/(h ft2 ?F)To = 200?FT?? = 80?FUse a grid spacing...
-
Using your results from Problems P5.18 and P5.7, calculate ÎS, ÎS surroundings , and ÎS total for each step in the cycle and for the total Carnot cycle described in Figure 5.2....
-
Explain the difference between an error of commission and an error of principle.
-
List and briefly describe the major types of shoplifters.
-
If the hollow cylinder of Problem 66 is replaced with a solid sphere, will the minimum value of h increase, decrease, or remain the same? Once you think you know the answer and can explain why, redo...
-
Unlikely Corporation has a working interest in Lease A. As of 12/31/15, the lease had reserves as follows: Calculate DD&A for each quarter of 2016, assuming the following production, using both...
-
A gas-turbine power plant operates on the simple Brayton cycle between the pressure limits of 100 and 700 kPa. Air enters the compressor at 30C at a rate of 12.6 kg/s and leaves at 260C. A diesel...
-
on octtober 1 of the current year riley the sales manager of osborne corportation prepared a cash budget for oct, nov, dec. All of osbornes sales are made on account. August sales (actual) 40,000,...
-
Buildings in flood zones are difficult to insure by private insurers because the ideal requirements of an insurable risk are difficult to meet. a. Identify the ideal requirements of an insurable...
-
How can McDonalds cook 16 hamburgers in 45 seconds?
-
At the transition temperature of 95.4C, the enthalpy of transition from rhombic to monoclinic sulfur is 0.38 kJ mol 1 . a. Calculate the entropy of transition under these conditions. b. At its...
-
What is the expected value of a zero-coupon bond, that is: Under the following dynamics: What distribution does the zero-coupon bond follow? Consider the distribution of st rudu rudu. In addition...
-
Explains your leadership philosophy based on leadership models and theories of your choosing. What will be your leadership style? Which traits do you believe will inspire others? What qualities do...
-
Today is January 1, 2022. Archie and Elaine Peyton have come to you, a financial planner, for help in developing a plan to accomplish their financial goals. From your initial meeting together, you...
-
Once you determine an individual's leadership readiness, giving them opportunities to expand their leadership ability is essential. Rothwell (2010) suggested some powerful leadership development...
-
Your answer should be in the form of an essay. The essay should be well organized, that is, it has an introduction (5 marks) , body (90 marks) and conclusion (5 marks) . The introduction should...
-
The idea of leadership and the word "leader" has been used in political and business scholarship to denote both those in formal power positions (i.e., king, head of state, president, CEO), as well as...
-
Visit http://www.cbc.ca/news/health/simple-hospital-innovation-saves-time-money-1.1298578 to see an application of lean in a hospital. What happened in the hospital?
-
Determine the annual percentage yield for a loan that charges a monthly interest rate of 1.5% and compounds the interest monthly.
-
Find the angle between A and B if A = 3.00i + 2.00j + 1.00k and B = 1.00i + 2.00j + 3.00k.
-
A spherical object falling in a fluid has three forces acting on it: (1) The gravitational force, whose magnitude is F g = mg, where m is the mass of the object and g is the acceleration due to...
-
An object has a force on it given by F = (4.75 N)i + (7.00 N)j + (3.50 N)k. (a) Find the magnitude of the force. (b) Find the projection of the force in the x-y plane. That is, find the vector in the...
-
Consider how Star Valley, a popular ski resort, could use capital budgeting to decide whether the $8 million Blizzard Park Lodge expansion would be a good investment. Assur conce assur Click the icon...
-
Selected T-accounts of Moore Company are given below for the just completed year: Raw Materials Debit Credit Balance 1/1 28,000 Credits Debits 146,000 Balance 12/31 38,000 Manufacturing Overhead...
-
Your company has total receivables outstanding at 31 December 2020 of $20,000. You think that about 2% of these balances will not be collected and wish to make an appropriate allowance. You have not...

Study smarter with the SolutionInn App


