What can you conclude about the ratio of fugacity to pressure for N 2 ,H 2 ,
Question:
Figure 7.10
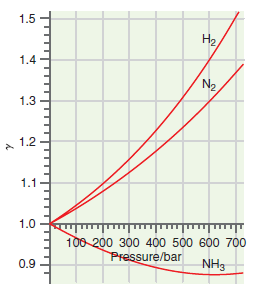
Transcribed Image Text:
1.5 H2, 1.4 N2 1.3 1.2 1.1 1.0 100 200 300 400 500 600 700 Pressure/bar 0.9 NH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
F P for ...View the full answer

Answered By

Sarfraz gull
have strong entrepreneurial and analytical skills which ensure quality tutoring and mentoring in your international business and management disciplines. Over last 3 years, I have expertise in the areas of Financial Planning, Business Management, Accounting, Finance, Corporate Finance, International Business, Human Resource Management, Entrepreneurship, Marketing, E-commerce, Social Media Marketing, and Supply Chain Management.
Over the years, I have been working as a business tutor and mentor for more than 3 years. Apart from tutoring online I have rich experience of working in multinational. I have worked on business management to project management.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
If A = B, what can you conclude about the components of A and B?
-
You are interested in discerning the role of Ï factors in prokaryotic transcription, using a system of purified core polymerase, a DNA template, a Ï factor, and labeled nucleotides...
-
What can you conclude about the interaction between ions in an electrolyte solution if the mean ionic activity coefficient is greater than one?
-
A person who is 6 feet and 3 inches tall and weighs 185 pound force (lbf) is driving a car at a speed of 65 miles per hour over a distance of 25 miles between two cities. The outside air temperature...
-
Shady Lane's Income tax payable account decreased from $14.3 million to $11.7 million during 2016. If its Income tax expense was $80.2 million, what was shown a an operating cash flow under the...
-
Let A be a fixed positive constant, and define the function f(x) by f(x) = 1/2 e- x if x > 0 and f(x) = 1/2 ex if x < 0. (a) Verify that f(x) is a pdf. (b) If A" is a random variable with pdf given...
-
Describe the term cost driver in relation to ABC. Give an example of a cost driver in events, hospitality and tourism.
-
Comparative financial statements for Weller Corporation, a merchandising company, for the year ending December 31 appear below. The company did not issue any new common stock during the year. A total...
-
Which of the following clients would not have a Schedule C? All of them received a Form 1099-K.
-
In 2016, taxicabs in Los Angeles charged an initial fee of $2.85 plus $2.70 per mile. In equation form, Fare Fare = 2.85 + 2.7(miles). At the end of a month, a businessman collects all his taxicab...
-
Predict the major product of the reaction between 1-butanol and: (a) PBr 3 (b) SOCl 2 , py (c) HCl, ZnCl 2 (d) H 2 SO 4 , heat (e) PCC, CH 2 Cl 2 (f ) Na 2 Cr 2 O 7 , H 2 SO 4 , H 2 O (g) Li (h) NaH...
-
A system consisting of 82.5 g of liquid water at 300. K is heated using an immersion heater at a constant pressure of 1.00 bar. If a current of 1.75 A passes through the 25.0 ohm resistor for 100. s,...
-
Incomplete financial statements for Pepper Industries follow: Pepper Industries Income Statement For the Year Ended March 31 The following additional information is available about the company: a....
-
Database store concepts question about blocking 1. [10] La Trobe University has approximately 20,000 active students. On the average, a student data will require 500 characters. The student table is...
-
Martin Corporation sells component parts for the electronics industry.Martin Corporation currently sells 160,000 units per year at a price of $6.50 per unit; its variable cost is $4.00 per unit; and...
-
The following data are available for the most recent year of operations for Slacker & Sons. The revenue portion of the sales activity variance is $279,000 F Master budget based on actual sales of...
-
treated with HCI. Draw the Products that would result if the following compounds were
-
The Blank Manufacturing Company Ltd. Consists of four production departments and two service departments.For the month of September the direct departmental expenses were as follows: Production...
-
The magnetic resonance imaging (MRI) unit of St Francis hospital has 2 MRI machines and sees two kinds of patients, regular patients and contrast patients. The regular patients dont need any special...
-
Which of the following streaming TV devices does not involve use of a remote controller? A) Google Chromecast B) Apple TV C) Amazon Fire TV D) Roku
-
Which of the molecules in Exercises 12.9b and 12.10b can be? (a) Polar, (b) Chiral?
-
Consider the C3Yion NO;. Is there any orbital of the central N atom that can have a nonzero overlap with the combination 2pz (A) pz (B) pz (C) of the three O atoms (with z perpendicular to the...
-
The CIO2 molecule (which belongs to the group C, 2v) was trapped in a solid. Its ground state is known to be BJ Light polarized parallel to the y-axis (parallel to the 00 separation) excited the...
-
Mint, now part of Intuit, entered the market in 2005 with comprehensive online money management systems that by aggregating financial information from online bank and credit card accounts tell users...
-
Which benefit has federal regulations that directly tie the physician's benefit to the employees' benefit (if offered to physician must also be offered to employee)? a. Flexible Spending Account...
-
Which of the following is a prohibited act under Chapter 673? Question 20Select one: a. To use a suspended license b. To prepare a fraudulent return c. To use the license of another person d. All of...

Study smarter with the SolutionInn App


