Each of the following answers indicates the quantum mechanical states of two electrons, A and B. Which
Question:
Each of the following answers indicates the quantum mechanical states of two electrons, A and B. Which pair of states could not describe two of the electrons in a multiple-electron atom?
a.
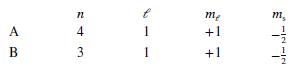
b.
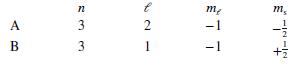
c.
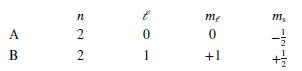
d.
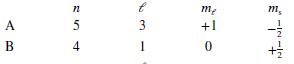
e.
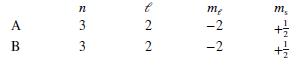
Transcribed Image Text:
me m, 4 1 +1 В 1 +1 AB
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
e The Pauli exclusion princ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Physics
ISBN: 9781119539636
11th Edition
Authors: John D. Cutnell, Kenneth W. Johnson, David Young, Shane Stadler
Question Posted:
Students also viewed these Sciences questions
-
Each of the following situations involves possible violations of the AICPA Code of Professional Conduct. For each situation, state whether it is a violation of the Code. In those cases in which it is...
-
Each of the following situations has an internal control weakness. a. Linda Grable has been your trusted employee for 30 years. She performs all cash-handling and accounting duties. Linda just...
-
The following tables show the domestic supply and demand schedules for bushels of flaxseed (used as an edible oil and a nutrition supplement) in the United States and Kazakhstan, with prices measured...
-
Write a reflective paper about Disability Network West Michigan
-
Write an equation for a function that has the shape of y = x2, but reflected across the x-axis and shifted left 2 units and up 3 units.
-
Derive the vant Hoff equation given in problem 15.28, using Eqs.15.12 and 15.15. Note: the d (g-/T) at constant P for each component can be expressed using the relations in Eqs. 13.18 and 13.19.
-
Question: When a man robbed a convenience store, he locked the clerk in the bathroom and took her purse. Forty minutes later, he used her ATM card to withdraw $500 from a bank and her checkbook to...
-
Klumb Inc. uses a periodic inventory system. Its records show the following for the month of May, in which 74 units were sold. InstructionsCalculate the ending inventory at May 31 using the (a) FIFO,...
-
30 Points) Highland's TV-Radio Store must determine how many TVs and radios to keep in stock. A TV requires 10 sq. ft. of floor-space, whereas a radio requires 4 sq ft. A total of 200 sq. ft. of f...
-
A simple model is sometimes used in order to illustrate the production-scheduling maxim, "balance flow, not capacity." Consider a factory that consists of three workstations where each customer order...
-
The voltage across an X-ray tube is 35.0 kV. Suppose that the molybdenum (Z = 42) target in the X-ray tube is replaced by a silver (Z = 47) target. Determine (a) The tubes cutoff wavelength (b) The...
-
Which one of the following statements is false? (a) The orbits in the Bohr model have precise sizes, whereas in the quantum mechanical picture of the hydrogen atom they do not. (b) In the absence of...
-
Use the properties of infinite series to evaluate the following series. (0.2)* + (08)
-
A stock has a return on equity of 10.5% and a plowback ratio of 30%. What is the sustainable growth rate? Enter you answer as a percentage.
-
In pandemic era, many people are affected, including job loss, poverty, unemployment, deflation and so on. All of this are related to the demand for money/currency. Get the statistics/report related...
-
James Bond conducts a top-down analysis of a fund and finds that the fund has a positive alpha of 1% per annum, a tracking error of 9% per annum (TE of less than 2% per annum should be considered as...
-
Propose a solution to the following scenario using each of the five ethical theories presented in this module. Explain how your solution aligns with the major ideas within each theory. Scenario:...
-
In the essay entitled "The Rising Cost of Not Going to College" by the Pew Research Center it is clear that the organization has concluded that going to college is a good investment for one's future....
-
Using the four Ps, discuss how the Apple iBeacon will create value for customers?
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Solid molecular hydrogen for H 2 one finds from measurements on the gas that the Lenard-Jones parameters are = 50 x 10 -16 erg and = 2.96 A. Find the cohesive energy in kJ per mole of H 2 ; do the...
-
Possibility of ionic crystals R + R Imagine a crystal that exploits for binding the coulomb attraction of the positive and negative ions of the same atom or molecule R. This is believed to occur...
-
Linear ionic crystal Consider a line of 2N ions of alternating charge q with a repulsive potential energy A/R n between nearest neighbors. (a) Show that at the equilibrium separation (CGS) U (R 0 )...
-
Meyer & Smith is a full-service technology company. They provide equipment, installation services as well as training. Customers can purchase any product or service separately or as a bundled...
-
Materials are entered at the beginning of the process. Conversion costs are incurred uniformly throughout the process. (a) Determine the equivalent units of production for (1) materials and (2)...
-
CoolSystems manufactures an optical switch that it uses in its final product. CoolSystems incured the following manufacturing costs when it produced 65,000 units last year (Click the icon to view the...

Study smarter with the SolutionInn App


