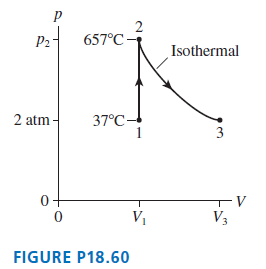
8.0 g of helium gas follows the process 1 ?? 2 ?? 3 shown in FIGURE P18.60.
Question:
8.0 g of helium gas follows the process 1 ?? 2 ?? 3 shown in FIGURE P18.60. Find the values of V1, V3, p2, and T3.
Transcribed Image Text:
P2- 657°C - Isothermal 2 atm - 37°C- 3 0+ -v V1 V3 FIGURE P18.60
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
Model Assume that the helium gas is an ideal gas Visualize Process 1 2 is isochoric proc...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Consider the following gas samples: 4.0 g of hydrogen gas, 4.0 g of helium gas, 1.0 mole of fluorine gas, 44.0 g of carbon dioxide gas, and 146 g of sulfur hexafluoride gas. Arrange the gas samples...
-
A monatomic gas follows the process 1 2 3 shown in Figure EX19.32. How much heat is needed for (a) Process 1 2 (b) Process 2 3? p (atm) 3- 2- 100C isotherm 3 -V (cm) 300 100 200 FIGURE EX19.32
-
Consider a container like that shown in Figure 20.12, with n 1 moles of a monatomic gas on one side and n 2 moles of a diatomic gas on the other. The monatomic gas has initial temperature T 1i ....
-
Court Casuals has 100,000 shares of common stock outstanding as of the beginning of the year and has the following transactions affecting stockholders' equity during the year. May 18 Issues 25,000...
-
Mauve, Inc., uses a two- to six-year graded vesting approach in its retirement plan. Calculate the nonforfeitable percentage for each of the following participants based upon the years of service...
-
A helium atom is in a finite potential well. The atoms energy is 1.0 eV below U 0 . What is the atoms penetration distance into the classically forbidden region?
-
(a) Josh Avers believes that the analysis ol linancial statements is directed at two characteristics of a company: liquidity and probability. Is Josh collect? Explain, (b) Are short-term creditors,...
-
1. Which forces facing Bivens supported change and which resisted change? 2. Did she use an economic or an organizational development approach to create change? Was it effective? 3. Using the...
-
Question 9 A company is considering expanding their production capabilities with a new machine that costs $44,000 and has a projected lifespan of 6 years. They estimate the increased production will...
-
Heat is conducted along a metal rod positioned between two fixed temperature walls. Aside from conduction, heat is transferred between the rod and the surrounding air by convection. Based on a heat...
-
Texas Instruments (TI) designs and manufactures semiconductor products for use in computers, telecommunications equipment, automobiles, and other electronics-based products. The manufacturing of...
-
JCPenney operates a chain of retail department stores, selling apparel, shoes, jewelry, and home furnishings. It also offers most of its products through catalog distribution. During fiscal Year 5,...
-
Important quiz hint: Know the difference between reinforcer and reinforcement so well that you dont mess it up, even in the heat of a quiz, now or ever.
-
1) Explain the term efficient portfolio. 2)How are the security market line and the capital asset pricing model related? 3)Assume you've drawn the efficient frontier and the risk-free rate exists....
-
Explain the importance of enzyme compartmentalization within eukaryotic cells and its significance for metabolic regulation and cellular signaling .
-
Sweet Catering completed the following selected transactions during May 2016: May 1: Prepaid rent for three months, $3,000 . May 5: Received and paid electricity bill, $170 May 9: Received cash for...
-
At the end of the current year, Accounts Receivable has a balance of $880,000, Allowance for Doubtful Accounts has a credit balance of $8,000, and sales for the year total $3,960,000. Bad debt...
-
As a coffee enthusiast, you wish to keep your coffee as hot as possible for as long as possible, so you decide to determine the rate of heat loss of your favority coffee mug. The mug is a cylinder...
-
When is the suspension of an employee a reasonable course of conduct?
-
Consider the advantages and disadvantages of extending property rights so that everyone would have the right to prevent people imposing any costs on them whatsoever (or charging them to do so).
-
In which of the four basic interactions does an electron participate? A neutrino? A proton? A photon?
-
What is a quark? How many different types of quarks are now known? What are some of the basic properties that distinguish these quarks?
-
Describe the kinds of evidence that have led scientists to conclude that quarks exist.
-
Can you please answer the following question in two paragraphs, 5 sentences each paragraph, and in your own words. 1. Can you explain the obstacles that the LGBTQ community have faced before and now...
-
Recording a Bond with a Discount or Premium Sold a bond with a face value of $10,000,000 for $9,683,000. What is the journal entry to record the bond? The bond has a stated rate of 9% and is payable...
-
Summerlin Company budgeted 4,800 pounds of material costing $5.00 per pound to produce 2,600 units. The company actually used 5,300 pounds that cost $5.10 per pound to produce 2,600 units. What is...

Study smarter with the SolutionInn App


