a. Calculate the de Broglie wavelength of the electron in the n = 1, 2, and 3
Question:
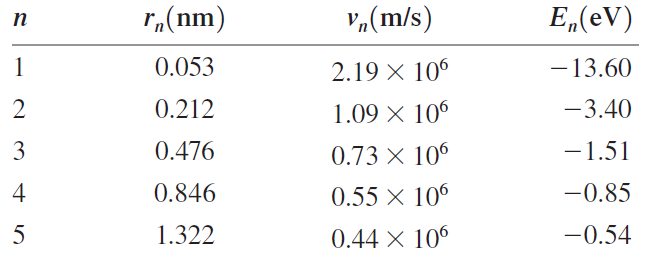 b. Show numerically that the circumference of the orbit for each of these stationary states is exactly equal to n de Broglie wavelengths.
b. Show numerically that the circumference of the orbit for each of these stationary states is exactly equal to n de Broglie wavelengths.
c. Sketch the de Broglie standing wave for the n = 3 orbit
Transcribed Image Text:
r„(nm) "¼(m/s) E„(eV) п 0.053 -13.60 2.19 X 106 2 0.212 -3.40 1.09 X 106 3 0.476 -1.51 0.73 × 106 4 0.846 0.55 × 106 -0.85 5 1.322 -0.54 0.44 X 10°
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
Solve a Using the data in Table 382 the wavelength of the electron in the n ...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A neutron in a reactor has kinetic energy of about 0.02 eV. Calculate the de Broglie wavelength of this neutron from Equation 17-12, where mc2 = 940 MeV is the rest energy of the neutron. hc 1240 eV...
-
We saw in Impact I8.1 that electron microscopes can obtain images with several hundredfold higher resolution than optical microscopes because of the short wavelength obtainable from a beam of...
-
Why Don't We Diffract? (a) Calculate the de Broglie wavelength of a typical person walking through a doorway. Make reasonable approximations for the necessary quantities. (b) Will the person in part...
-
Kim and Kanye have been dating for years and are now thinking about getting married. As a financially sophisticated couple, they want to think through the tax implications of their potential union....
-
Astronomers view light coming from distant galaxies moving away from Earth at speeds greater than 10% of the speed of light. How fast does this light meet the telescopes of the astronomers?
-
A childs grandparents purchase a $10,000 bond fund that matures in 18 years to be used for her college education. The bond fund pays 4% interest compounded semiannually. How much will the bond fund...
-
Why is it necessary for financial reporting to be subject to (a) mandatory control and (b) statutory control?
-
Natalie has prepared the balance sheet and income statement of Cookie & Coffee Creations Inc. and would like you to prepare the statement of cash flows. Go to the books companion website,...
-
Required: Determine the costs assigned to the December 31 ending inventory based on the FIFO method. Perpetual FIFO: Goods Purchased Cost of Goods Sold Inventory Balance # of Date Cost Per # of Units...
-
Leather Works is a family-owned maker of leather travel bags and briefcases located in the northeastern part of the United States. Foreign competition has forced its owner, Heather Gray, to explore...
-
a. What quantum number of the hydrogen atom comes closest to giving a 100-nm-diameter electron orbit? b. What are the electrons speed and energy in this state?
-
What is the radius of a hydrogen atom whose electron moves at 7.3 10 5 m/s?
-
In tropical climates, the water near the surface of the ocean remains warm throughout the year as a result of solar energy absorption. In the deeper parts of the ocean, however, the water remains at...
-
The confirmation of customers accounts receivable rarely provides reliable evidence about the completeness assertion because a. Many customers merely sign and return the confirmation without...
-
To determine whether internal controls operated effectively to minimize errors of failure to invoice a shipment, the auditor would select a sample of transactions from the population represented by...
-
What is batch processing?
-
Briefly describe six reasons why the auditor's approach to obtaining an understanding of internal control is different when a computer is used rather than manual processing.
-
A manufacturing client received a substantial amount of goods returned during the last month of the fiscal year and the first month after year-end. The client recorded the returns when credit memos...
-
You are buying a new Porsche Boxter, priced at $65,200. You will pay $4,700 now and the rest monthly, in a four-year loan. All rates are APRs. The automobile dealership is offering a sales promotion...
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
Pipes often contain solid residue that adheres to the inner wall of the pipe. If these pipes are not properly cleaned before use, then the flow of water through the pipe will slowly dissolve the...
-
Natural gas from a hydraulic fracturing process contains hydrogen sulfide (H 2 S) gas that will be removed by absorption into a specialty solvent through an interphase mass transfer process. In the...
-
A 15-cm-OD pipe is buried with its centerline 1.25 m below the surface of the ground (k of soil is 0.35 W/(m K)). An oil having a density of 800 kg/m 3 and a specific heat of 2.1 kJ/(kg K) flows in...
-
The following selected transactions are from Mitchell Company. Year 1 December 16 Accepted a $20,400, 60-day, 12% note in granting Kathy Perry a time extension on his past-due account receivable....
-
Highland Company produces a lightweight backpack that is popular with college students. Standard variable costs relating to a single backpack are given below: Standard Quantity or Hours Standard...
-
Way Cool produces two different models of air conditioners. The company produces the mechanical systems in its components department. The mechanical systems are combined with the housing assembly in...

Study smarter with the SolutionInn App


