A gas with an initial temperature of 900°C undergoes the process shown in FIGURE EX18.36. a. What
Question:
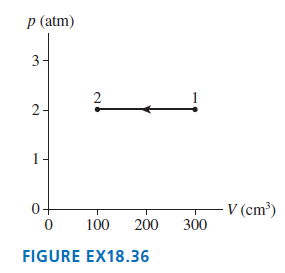 a. What type of process is this?
a. What type of process is this?
b. What is the final temperature in °C?
c. How many moles of gas are there?
Transcribed Image Text:
p (atm) 3- 2 2- 1- V (cm³) 300 0+ 100 200 FIGURE EX18.36
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
Model Assume that the gas is ideal Solve a Because the process i...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Five moles of an ideal monatomic gas with an initial temperature of 127C expand and, in the process, absorb 1200 J of heat and do 2100 J of work. What is the final temperature of the gas?
-
A 0.6-cm-thick flat plate of stainless steel, 7.5 cm wide and 0.3 m long, is immersed horizontally at an initial temperature of 980?C in a large water bath at 100?C and at atmospheric pressure....
-
A plastic tube of 7.6-cm ID and 1.27 cm wall thickness having a thermal conductivity of 1.7 W/(m K), a density of 2400 kg/m3, and a specific heat of 1675 J/(kg K) is cooled from an initial...
-
ABC Company produces mode trains. During the month of July, it produced 4,000 trains. The actual labor hours were 8 hours per train. Its standard labor hours are 10 hours per train. The standard...
-
What purpose is served by the check-the-box Regulations?
-
The answer to Exercise 19 part (d) is needed for this exercise. A geologist counts 64 emitted particles in one minute from a certain radioactive rock. a. Find a 95% confidence interval for the rate...
-
The chapter stresses the importance of comparing an individual company's financial ratios to industry norms. Robert Morris Associates (RMA), a national association of bank loan and credit officers,...
-
You are creating a customer database for the Lehigh Valley IronPigs minor league baseball team. Draw a project network given the information below. Complete the forward and backward pass, compute...
-
If the probability that an event will occur is odds in favor of the event occurring are The odds are (Simplify your answer.) 1 8 then the probability that the event will not occur is and th 9' 9'
-
Steeley Associates, Inc., a property development firm, purchased an old house near the town square in Concord Falls, where State University is located. The old house was built in the mid-1800s, and...
-
0.0040 mol of gas undergoes the process shown in FIGURE EX18.35. a. What type of process is this? b. What are the initial and final temperatures in °C? p (atm) 3- 2- V (cm) 300 0+ 100 200 FIGURE...
-
0.020 mol of gas undergoes the process shown in FIGURE EX18.37. a. What type of process is this? b. What is the final temperature in °C? c. What is the final volume V 2 ? p (atm) 3- 2- 1- V (cm)...
-
Is the United States the nation with the most government debt? If not, who is?
-
Bruce is considering the purchase of a restaurant named Hard Rock Hollywood. With the help of his accountant, Bruce projects the net cash flows (cash inflows less cash outflows) from the restaurant...
-
Use multiplication: 6. (secx+1)(secx-1)= tan x 9. cos x(secx-tanx)=1-sin x 7. sin x(cscx - sin x) = cos x 10. cotx(secx-cos x)=sin x 8. (sin x cos x)=1-2 sin x cos x
-
The drawing shows an electron entering the lower left side of a parallel plate capacitor and exiting at the upper right side. The initial speed of the electron is 1.61 106 m/s. The capacitor is 2.00...
-
Give an example of each of the following, or provide an argument for why the request is impossible. Assume that all of the functions discussed below are defined on all of R. (i) A function that is...
-
Sarasota Industries sells three different sets of sportswear. Sleek sells for $48 and has unit variable costs of $36; Smooth sells for $68 and has unit variable costs of $48; Potent sells for $78 and...
-
Name and describe two types of union security clauses and identify their common purpose in a collective agreement.
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
When a tidal barrage follows the water release profile shown in Figure 31.27 it only captures a fraction of the energy (31.35) available in the water stored at high tide. The discharge is indicated...
-
Consider an idealized tidal reservoir of surface area A = 30 km 2 that fills and drains through a narrow channel of width w = 60 m and depth d = 20 m. Assume that the tidal range is h = 2.5 m. For...
-
Estimate the total power of the Gulf Stream in the Florida Straits, given that the mass flux is roughly 30 Sv. Take the speed of the flow to be 4 m/s.
-
Cost of Production Report Hana Coffee Company roasts and packs coffee beans. The process begins by placing coffee beans into the Roasting Department. From the Roasting Department, coffee beans are...
-
More info 1. Suppose you invest a sum of $2,000 in an interest-bearing account at the rate of 10% per year. What will the investment be worth six years from now? 2. How much would you need to invest...
-
Schriever Corporation is an oil well service company that measures its output by the number of wells serviced. The company has provided the following fixed and variable cost estimates that it uses...

Study smarter with the SolutionInn App


