A heat engine using a diatomic gas follows the cycle shown in FIGURE P21.56. Its temperature at
Question:
a. Determine Ws, Q, and ΔEthfor each of the three processes in this cycle. Display your results in a table.
b. What is the thermal efficiency of this heat engine?
c. What is the power output of the engine if it runs at 500 rpm?
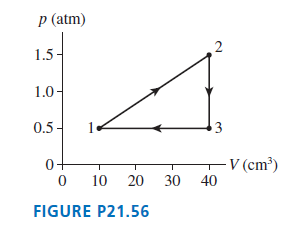
Transcribed Image Text:
p (atm) 2 1.5- 1.0- 0.5 - - V (cm³) 40 10 20 30 FIGURE P21.56 3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
Model The heat engine follows a closed cycle For a diatomic gas C V 52 R and C P 72 R Solve ...View the full answer

Answered By

Deepak Pal
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students. Areas of interest: Business, accounting, Project management, sociology, technology, computers, English, linguistics, media, philosophy, political science, statistics, data science, Excel, psychology, art, history, health education, gender studies, cultural studies, ethics, religion. I am also decent with math(s) & Programming. If you have a project you think I can take on, please feel welcome to invite me, and I'm going to check it out!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A heat engine using a diatomic ideal gas goes through the following closed cycle: Isothermal compression until the volume is halved. Isobaric expansion until the volume is restored to its initial...
-
A heat engine uses a diatomic gas in a Brayton cycle. What is the engines thermal efficiency if the gas volume is halved during the adiabatic compression?
-
Consider a heat engine that is not reversible. The engine uses 1.000 mol of a diatomic ideal gas. In the first step (A) there is a constant temperature expansion while in contact with a warm...
-
2. LIMIT COMPARISON TEST: SPECIAL CASES The Limit Comparison Test is most often used when the limit L= lim ak lies in the interval (0, oo). In the case where L is zero or infinity, we can still...
-
Evaluate the competitive strategies for firms in foreign markets: a. Niche market exporting b. Licensing and contract manufacturing c. Franchising d. Joint ventures e. Wholly owned subsidiaries
-
Should Jacqueline and Keith cash out the excess equity that they have built up? Assume money market rates are 2.15%.
-
Quick Electronics Inc. produces and sells two models of pocket calculators, XQ-103 and XQ-104. The calculators sell for $12 and $20, respectively. Because of the in- tense competition Quick faces,...
-
Kase Company can invest in each of three cheese-making projects: C1, C2, and C3. Each project requires an initial investment of $190,000 and would yield the following annual cash flows. (1) Assuming...
-
Solve the system by back substitution. -2x-4y-z-3w= 1 -y-6z-2w=-8 -5z-5w=-10 -5w=-15 The solution set is {} (Type an integer or a simplified fraction.)
-
Quality Air Conditioning manufactures three home air conditioners: an economy model, a standard model, and a deluxe model. The profits per unit are $63, $95, and $135, respectively. The production...
-
A heat engine using 1.0 mol of a monatomic gas follows the cycle shown in FIGURE P21.55. 3750 J of heat energy is transferred to the gas during process 1 2. a. Determine W s , Q, and ÎE th for...
-
FIGURE P21.57 shows the cycle for a heat engine that uses a gas having Ï = 1.25. The initial temperature is T 1 = 300 K, and this engine operates at 20 cycles per second. a. What is the power...
-
You own a share of Costco stock. You are worried that its price will fall and would like to insure yourself against this possibility. How can you purchase insurance against a fall in the price of the...
-
Robotix Inc. purchased a patent for $20,000 on January 1. The patent is good for 10 years, but Robotix plans to produce and sell products using that patent technology for at least 15 years. c) use...
-
3 Quanta forecasts this year's cash flow will be $750,000.00. Justin estimates the appropriate discount rate for its cash flows is 3.35%, but his colleague suggests it is 55 bps higher. If they agree...
-
You deposit $3000 in an account that pays 8 % interest compounded semiannually. After 5 years, the interest rate is increased to 8.44 % compounded quarterly. What will be the value of the account...
-
2:3-24 Today's Electronics specializes in manufacturing modern electronic components. It also builds the 20equipment that produces the components. Phyl- lis Weinberger, who is responsible for...
-
The Lucy V . Company, has budgeted sales in units for the next five months as follows:JuneJuly 8 , 2 0 0 units 6 , 3 0 0 unitsAugust 4 , 7 0 0 unitsSeptember 3 , 6 0 0 unitsOctober 4 0 0 0 unitsPast...
-
Verizon Communications Inc. (VZ) is a major telecommunications company in the United States. Two recent balance sheets for Verizon disclosed the following information regarding fixed assets: Verizons...
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
Two masses are separated by a distance r. If this distance is doubled, is the force of interaction between the two masses doubled, halved, or changed by some other amount? Explain.
-
A painter depicts a portion of the night sky as shown in the diagram below, showing the stars and a crescent moon. Is this view possible? Explain. Q24 Diagram
-
At what times during the day or night would you expect the new moon to rise and set? Explain.
-
In developing a budget, which of the following should be considered? I. Balance sheet II. Chart of accounts III. Expenses from the current/past year IV. Purchasing contracts V. Physician compensation...
-
JOIICS SUIT SHOPSales BudgetQuarter 1Quarter 2Quarter 3Expected Sales Units150175Sales Price per Unit$250250$250otal Sales Revenue37,500$ JOIICS SUIT SHOPSales BudgetQuarter 1Quarter 2Quarter...
-
Which of the following statements is true concerning foreign income tax paid? Question 39Select one: a. If a taxpayer takes a subtraction on their Oregon return, then the subtraction is included as...

Study smarter with the SolutionInn App


