Draw a first-law bar chart (see Figure 19.12) for the gas process in Figure EX19.6. Figure 19.12
Question:
Figure 19.12
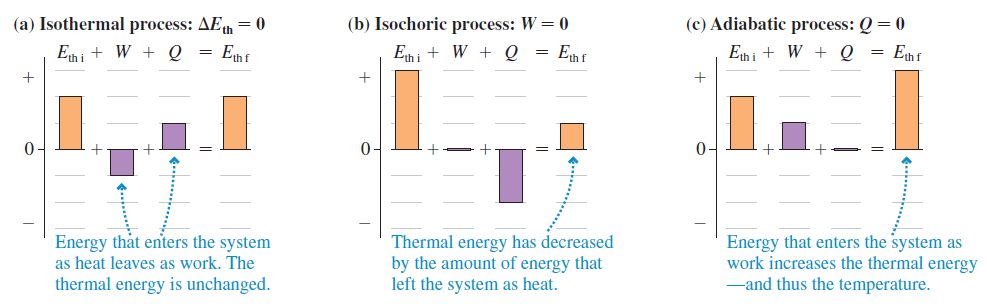
Transcribed Image Text:
FIGURE EX19.6 (a) Isothermal process: AEh = 0 (b) Isochoric process: W = 0 (c) Adiabatic process: Q = 0 Eth i + W + Q = Enf En i + W + Q Enf Ehi + W + Q = Enf 0- Energy that enters the system as heat leaves as work. The thermal energy is unchanged. Thermal energy has decreased by the amount of energy that Energy that enters the system as work increases the thermal energy -and thus the temperature. left the system as heat.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)
Visualize Solve Because this is an isobaric process W pdV pV f V i Since V f ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Figure 2.1 shows a bar chart and a pie chart for how people paid for purchases and other transactions (see page 47). a. Create an exploded pie chart, a cone chart, or a pyramid chart that shows how...
-
Figure 2.1 shows a bar chart and a pie chart for the main reason young adults shop online. a. Create an exploded pie chart, a doughnut chart, a cone chart, or a pyramid chart that shows the main...
-
Figure 2.1 shows a bar chart and a pie chart for what bosses demand during vacation time . a. Create an exploded pie chart, a doughnut chart, a cone chart, or a pyramid chart that shows what bosses...
-
According to Electronic Designs 2012 Engineering Salary Survey, the mean base salary of a software engineering manager is $126,417the highest mean among all types of engineers. In contrast, a...
-
Excited hydrogen and excited helium atoms both radiate light as they jump down to the n = 1, = 0, m = 0 state. Why do the two elements have very different emission spectra?
-
Determine the compensation capacitor for the two-stage op amp in the example in Section 9.4.3 that gives a 60 phase margin.
-
Toyota Exports to the United Kingdom. Toyota manufactures in Japan most of the vehicles it sells in the United Kingdom. The base platform for the Toyota Tundra truck line is 1,650,000. The spot rate...
-
The March 31, 2014, adjusted trial balance for sopik Refrigeration Repairs is shown below with accounts in alphabetical order. 1 $5,000 of the long-term note payable is due during the year ended...
-
ship is sailing east. At one point, the bearing of a submerged rock is 4820'. After the ship has sailed 14.5 mi, the bearing of the rock has become 30640'. Find the distance of the ship from the rock...
-
Consider the following I/O scenarios on a single-user PC. a. A mouse used with a graphical user interface b. A tape drive on a multitasking operating system (assume no device preallocation is...
-
Why is the molar specific heat of a gas at constant pressure larger than the molar specific heat at constant volume?
-
You throw a baseball straight up in the air so that it rises to a maximum height much greater than your height. Is the magnitude of the balls acceleration greater while it is being thrown or after it...
-
Presented below are the financial statements of Helwany Company. Additional data:1. Depreciation expense was $17,500.2. Dividends declared and paid were $20,000.3. During the year equipment was sold...
-
Find the solution of the exponential equation, correct to four decimal places. e-3x=9 x=
-
What is a potential disadvantage of an IDS?
-
1. Explain the objective of the capital asset pricing model 2. Explain the objective of the calculation of the break-even level per units/ revenues 3. When would you use the divisional WACC instead...
-
What should my journal entry's be to record the admission of morris based on the following information a company has total partner equity of 776,000 which is made up of marks capital 608,000 and...
-
What link should a user with Sign/Submit access select from the sidebar menu when preparing to submit a revised subapplication?
-
Havel Robotics Company (a U.S.-based firm) exports 25,000 industrial robots per year to China under an agreement that covers a 5-year period. In China, the robots are sold for the RMB (Chinese...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
What kind of charge does an object acquire when electrons are stripped from it?
-
Distinguish between dc and ac.
-
Which has the greater resistance, a thick wire or a thin wire of the same length?
-
The next eight questions cover income tax filing requirements for Oregon Larry is 26 years old and will use the single filing status. He was a full-year resident of Oregon and has no dependents. For...
-
As a junior associate for a CPA firm, you are asked to fomulate a plan for a client about audit services. The client has a passing knowledge of internal and external risks, the Sarbanes-Oxley Act,...
-
During the month of June?, Richardson?, Inc. had transactions that resulted in the following amounts being recorded in its Cash? account; (1) $ 1 comma 800 ?debit, (2) $ 450 ?credit, (3) $ 325...

Study smarter with the SolutionInn App


