FIGURE P20.50 shows the thermal energy of 0.14 mol of gas as a function of temperature. What
Question:
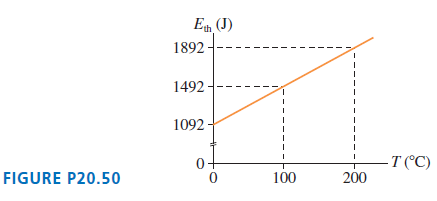
Transcribed Image Text:
E (J) 1892--- 1492 - 1092 T (°C) FIGURE P20.50 100 200
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (20 reviews)
Model Assume the gas is ideal so that Equation 2030 w...View the full answer

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
The energy from nuclear fission appears in the form of thermal energy-but the thermal energy of what?
-
A cylinder of nitrogen gas has a volume of 15,000 cm 3 and a pressure of 100 atm. a. What is the thermal energy of this gas at room temperature (20C)? b. What is the mean free path in the gas? c. The...
-
A metal tool is sharpened by being held against the rim of a wheel on a grinding machine by a force of 180 N. The frictional forces between the rim and the tool grind off small pieces of the tool....
-
2) WWW.myitlab.com is an example of a(n). O domain name O protocol prefix OURL omni box
-
In the 1990s many Nasdaq firms favored growth over profitability; in the 2000s the goal of profitability is displacing growth. How might each preference be explained?
-
1. Jamie Lee is beside herself knowing that the thieves had unauthorized use of her Debit/ATM card. What is Jamies financial responsibility for the unauthorized use? 2. What would have been Jamie...
-
For Advent Company, the budgeted cost for one unit of product is direct materials \($10\), direct labor \($20\), and manufacturing overhead 80% of direct labor cost. If 25,000 units are expected to...
-
Sports Drinks, Inc. began business in 2013 selling bottles of a thirst- quenching drink. Production for the first year was 104,000 bottles, and sales were 98,000 bottles. The selling price per bottle...
-
7. An engineer makes different-size samples of a new material. The table shows volumes and their related masses. a. What would be the mass of 100 cubic centimeters of the material? 3000 b. What would...
-
If collective bargaining and the grievance procedures have not been able to settle a current labor dispute, what tactics would you and other employees adopt? Which tactics would be best for which...
-
A 10 cm 10 cm 10 cm box contains 0.010 mol of nitrogen at 20C. What is the rate of collisions (collisions/s) on one wall of the box?
-
A 100 cm 3 box contains helium at a pressure of 2.0 atm and a temperature of 100C. It is placed in thermal contact with a 200 cm 3 box containing argon at a pressure of 4.0 atm and a temperature of...
-
Robert J. Lucas owned Big Hill Acres, Inc. (BHA, Inc.) and Consolidated Investments, Inc. Through these companies, he acquired Big Hill Acres (BHA), a large parcel of land in Jackson County,...
-
5.96x10^24kg Therefore, the mass of the earth is =2.2 lbs). Is your answer close to 5.98 x 1024 kg? kilograms (1 kg 2.2 Assuming an average human has a mass of 70. Kg, how many humans would it take...
-
Anna has six identical bottles of apple juice. If the serving glass is 3/4 of a bottle, how many servings can Anna make out of the bottles? Show your solution using a picture. Then provide a formal...
-
Consider the two different project types below. For each project type, consider which areas of the Cost Management Plan would be most important, and explain your rationale for each in the space...
-
Identify some qualitative factors that should be considered when making managerial decisions ( I would like to see 3 - 5 factors ) . Please explain what they are, what they mean, and why they should...
-
Question 2. Manatee Splash is going from stregth to strength. Production is at 22,500/day. Daily demand is currently 15,000 units/day. It is forecast that daily demand will triple next year. 2.1 How...
-
The unadjusted trial balance of La Mesa Laundry at August 31, 20Y5, the end of the fiscal year, follows: The data needed to determine year-end adjustments are as follows: (a) Wages accrued but not...
-
Design and describe an application-level protocol to be used between an automatic teller machine and a bank's centralized computer. Your protocol should allow a user 's card and password to be...
-
Is chemistry the study of submicroscopic, microscopic, or macroscopic matter, or of all three? Defend your answer.
-
You combine 50 mL of water with 50 mL of purified alcohol and get a total of 98 mL of the mixture. Please explain how this occurs.
-
A cotton ball is dipped in alcohol and wiped across a tabletop. Explain what happens to the alcohol molecules deposited on the tabletop. Is this a physical or chemical change?
-
Nancy is 21 and a full-time college student and is blind. Nancy lives with her mother and is claimed as a dependent by her mother. Her earned income was $6,500 for the 2022 tax year. Using the...
-
Christopher Foley, an attorney, has a law corporation, Christopher Foley, Attorney, Inc., that began the year with total assets of $145,000, total liabilities of $70,000, and stockholders' equity of...
-
Which of the following statements is correct? Group of answer choices All of these answers are correct. A subscriber must take reasonable steps to offer users a receipt for all transactions, before...

Study smarter with the SolutionInn App


