FIGURE Q2.6 shows the position-versus-time graph for a moving object. At which lettered point or points: a.
Question:
a. Is the object moving the slowest?
b. Is the object moving the fastest?
c. Is the object at rest?
d. Is the object moving to the left?
Figure Q2.6
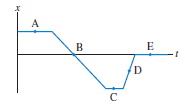
Transcribed Image Text:
А B E
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a B The object is still moving but the magnitude of the slope o...View the full answer

Answered By

Muhammad Ghyas Asif
It is my obligation to present efficient services to my clients by providing a work of quality, unique, competent and relevant. I hope you have confidence in me and assign me the order and i promise to follow all the instructions and keep time.
4.60+
109+ Reviews
203+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
Figure 2-36 is a position versus time graph for the motion of an object along the x axis. Consider the time interval from A to B (a) Is the object moving in the position or negative direction? (b) Is...
-
FIGURE Q2.7 shows the position-versus-time graph for a moving object. At which lettered point or points: (a) Is the object moving the fastest? (b) Is the object moving to the left? (c) Is the object...
-
FIGURE Q2.14 shows the velocity-versus-time graph for a moving object. At which lettered point or points: a. Is the object speeding up? b. Is the object slowing down? c. Is the object moving to the...
-
If any fi rms price of labor and capital each double, what will happen to the expansion path (i.e., locus of tangencies between the isoquants and isocost curves)? What will happen to the fi rms...
-
Determine the parameters of a parallel resonant circuit which has the following properties: Wo = 2Mrad/sec, BW = 20 krad/sec, and an impedance of 2000ohm.
-
An ideal-gas mixture whose apparent molar mass is 20 kg/kmol consists of N2 and three other gases. If the mole fraction of nitrogen is 0.55, its mass fraction is (a) 0.15 (b) 0.23 (c) 0.39 (d) 0.55...
-
Look at the annual report of an MNE. As you read about the product lines and geographical operations, what kind of organizational structure would you guess the company has?
-
Mike Moore's microbrewery is considering production of a new ale called Mike's Honey Harvest Brew. To produce this new offering he is considering two independent projects. Each of these projects has...
-
A magazine subscription service is having a contest in which the prize is $40,000. If the company receives 2 million entries, what is the expectation of the contest? $
-
Sam and John have been friends for 20 years. They met in college and worked together for 10 of the 20 years. During that time, each made a promise that if they won a lottery they would share the...
-
FIGURE Q2.5 shows a position-versus-time graph for the motion of objects A and B as they move along the same axis. a. At the instant t = 1 s, is the speed of A greater than, less than, or equal to...
-
FIGURE Q2.8 shows six frames from the motion diagrams of two moving cars, A and B. (a) Do the two cars ever have the same position at one instant of time? If so, in which frame number (or numbers)?...
-
You and a friend have decided to work jointly on a course project. Frankly, your friend is a less-than ideal partner. His skills as a researcher are such that he can review and outline only two...
-
You are auditing the rental income of a car hire company. Suggest the types of inherent and control risk which would be encountered in this type of audit and describe the analytical review and...
-
What are the various ways of sharing profits between partners, and why are they used?
-
What liability should the city report on government-wide f inancial statements at the end of Year 1? a. It depends on whether the employees work at governmental activities or business-type...
-
You are the auditor of a mail order company which sells clothes on an approval basis through a large number of customers spread across the country. Customers are given 30 days to pay for goods after...
-
Identify and quantify the costs that are relevant to a particular decision.
-
1. (a) What is the cash value of television A? Round your answer to the nearest dollar. (b) What is the cash value of television B? Round your answer to the nearest dollar. 2. (a) Given Michaels...
-
What do you think?
-
The electric output of a power plant is 750 MW. Cooling water flows through the power plant at the rate 1.0 10 8 L/h. The cooling water enters the plant at 16C and exits at 27C. What is the power...
-
Engineers testing the efficiency of an electric generator gradually vary the temperature of the hot steam used to power it while leaving the temperature of the cooling water at a constant 20C. They...
-
A heat engine using 1.0 mol of a monatomic gas follows the cycle shown in FIGURE P21.55. 3750 J of heat energy is transferred to the gas during process 1 2. a. Determine W s , Q, and ÎE th for...
-
During Heaton Company's first two years of operations, it reported absorption costing net operating income as follows: Sales (@ $62 per unit) Year 1 Year 2 $ Cost of goods sold (@ $43 per unit) Gross...
-
1. The numbers in cells B18 to B20 should be formulas that refer to B15. This allows the number of cases to be changed in cell 815 with the other cells changing automatically. Exce2. The formula in...
-
Based on the following transactions, answer the following questions. i. Purchased inventory with a cost of $28,300 on account. ii. Sales on account to customers totalled $54,700. iii. Payments made...

Study smarter with the SolutionInn App


