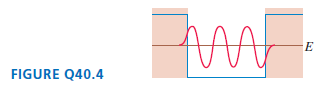
What is the quantum number of the particle in FIGURE Q40.4? How can you tell? E FIGURE
Question:
What is the quantum number of the particle in FIGURE Q40.4? How can you tell?
Transcribed Image Text:
E FIGURE Q40.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (13 reviews)
n 6 be...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Question Posted:
Students also viewed these Physics questions
-
A particle confined to a one-dimensional box has a ground-state energy of 0.4 eV. When irradiated with light of 206.7 nm it makes a transition to an excited state. When decaying from this excited...
-
What is the quantum number of an electron confined in a 3.0-nm-long one-dimensional box if the electrons de Broglie wavelength is 1.0 nm?
-
The contour representation of one of the orbital's for the n = 3 shell of a hydrogen atom is shown below. (a) What is the quantum number l for this orbital? (b) How do we label this orbital? (c) How...
-
A ball is thrown from the roof of a 1 0 0 m high building at a speed of 1 0 m / s making 3 0 angles with the horizontal. A child waiting on the ground at the door of the building falls into a coma...
-
Comment on this statement: "Oh, that beautiful red sunset is just the leftover colors that weren't scattered on their way through the atmosphere."
-
What does the dot member access operator indicate in the text System.Windows .Forms.Label?
-
Kemico Ltd manufactures a variety of chemicals which it supplies in bulk to industrial customers. Its factory at Gritborough employs 50 people, and its annual turnover in 1995 was 7.5M. During 1995,...
-
Vera Ernst is a licensed dentist. During the first month of the operation of her business, the following events and transactions occurred. April 1 Invested $20,000 cash in her business. 1 Hired a...
-
! Required information [The following information applies to the questions displayed below.] Cane Company manufactures two products called Alpha and Beta that sell for $180 and $145, respectively....
-
You are given the following details about Facts of Life plc. Breakdown of activities by percentage of total annual company turnover: Department stores: .................................... 30%...
-
FIGURE EX40.4 shows the wave function of an electron in a rigid box. The electron energy is 12.0 eV. What is the energy, in eV, of the next higher state? (x) FIGURE EX40.4
-
FIGURE EX40.5 is the probability density for an electron in a rigid box. What is the electron??s energy, in eV? O nm 0.45 nm FIGURE EX40.5
-
The value stored in an uninitialized variable is _________. a. Null b. Garbage c. Compost d. Its identifier
-
Describe the difference between proportionate liability and contributory negligence.
-
Critically explain the extent to which the audit expectations gap is a dynamic concept.
-
Define the term audit quality and outline how the FRC and audit report users can determine if audit quality has been achieved.
-
The audit expectations gap will never be closed. Discuss.
-
Comment on why the proposed changes to sales processes are necessary. Also, which changes would you recommend in terms of their: (a) selling activities? (b) sales value proposition?
-
List several aspects of the auditees information systems and IT that indicate the need for an IT specialist on the audit team.
-
The figure shows six containers, each of which is filled from the top. Assume that water is poured into the containers at a constant rate and each container is filled in 10 seconds. Assume also that...
-
a. Is there a highest temperature at which ice can exist? If so, what is it? If not, why not? b. Is there a lowest temperature at which water vapor can exist? If so, what is it? If not, why not?
-
What is the diameter of a copper sphere that has the same mass as a 10 cm 10 cm 10 cm cube of aluminum?
-
The cylinder in FIGURE Q18.4 is divided into two compartments by a frictionless piston that can slide back and forth. If the piston is in equilibrium, is the pressure on the left side greater than,...
-
Barnett Brass sells economy door knobs for $15 each. Unit product costs are as follows: Direct materials Direct labor Manufacturing overhead Total $ 3 2 6 $11 An order to purchase 4,000 units was...
-
Culver Inc. presented the following data. Net income $2,610,000 Preferred stock: 51,000 shares outstanding, $100 par, 7% cumulative, not convertible 5,100,000 Common stock: Shares outstanding 1/1...
-
Which of the following comments is true regarding Justin Paperny's comments during his presentation? A. Justin believes that the main reason he was sentenced to prison is because his actions were key...

Study smarter with the SolutionInn App


