Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here.
Question:
Calculate the pH of a 0.010 M solution of each amino acid in the form drawn here.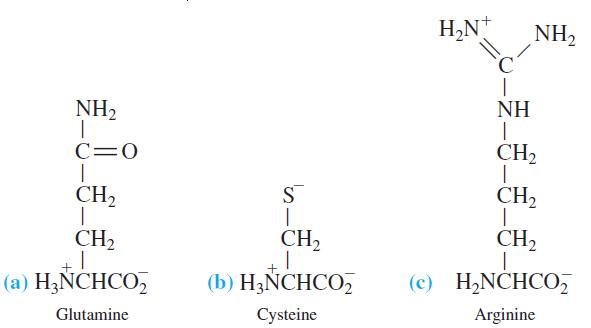
Transcribed Image Text:
H,N* NH2 NH2 NH C=0 CH2 CH2 S CH2 CH2 CH2 CH2 (a) H¿ÑCHCO, (b) H3ÑCHCO, (c) H,NCHCO, Glutamine Cysteine Arginine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
Amina acid PH PH of each Solution 0010 M The three forms of glutamine is as follows L...View the full answer

Answered By

Manoj Kumar Mahariya
Teaching experience-
1.As a lecture in Akash Carrer institute (since 2 year)
2.As a assistant lecturer in National Institute of technology,Punjab
3.Also school lecture of pcbm subject in Ravindra manorial senior sec.school, Rajasthan
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the pH of a solution prepared by (a) Dissolving 36.5 g of lactic acid in water and diluting to 500 mL. (b) Diluting 25.0 mL of the solution in (a) to 250 mL. (c) Diluting 10.0 mL of the...
-
Calculate the pH of a solution made by mixing 0.60 L of 0.10 M NH4Cl with 0.50 L of 0.10 M NaOH. Kb for NH3 is 1.8 10-5.
-
Calculate the pH of a solution made by mixing 7.52 mL of 4.9 10-2 M Ca(OH)2 with 22.5 mL of 0.11 M HCl.
-
The following information was taken from the annual manufacturing overhead cost budget of Granada Company. Variable manufacturing overhead costs .....$33,000 Fixed manufacturing overhead costs...
-
The GSS asked respondents to report their opinion on spanking as a method to discipline a child (SPANKING). Examine how respondents' attitudes toward spanking a child are associated with SEX, CLASS,...
-
Bill Youngdahl has been collecting data at the TU student grill. He has found that between 5:00 P.M. and 7:00 P.M., students arrive at the grill at a rate of 25 per hour (Poisson distributed) and...
-
'Drawings' is shown on the balance sheet as: a. An addition to the capital account b. A deduction from the capital account c. Part of current liabilities d. Part of current assets
-
A company buys a machine for $61,700 on January 1, 2008. The maintenance costs for the years 20082011 are as follows: 2008, $4,900; 2009, $4,700; 2010, $12,400 (includes $7,800 for cost of a new...
-
Walmart Inc. provides retail services using operating costing. In March 2034, the company incurs the following operating costs: Store Rent: $10 million Store Personnel Costs: $15 million Inventory...
-
Deepa Dalal opened a free-standing radiology clinic. She had anticipated that the costs for the radiological tests would be primarily fixed, but she found that costs increased with the number of...
-
Interpreting spectral data. The graph shows the 1H-nuclear magnetic resonance chemical shift of the H 4 proton on pyridine as a function of pH. Chemical shift is related to the environment of a...
-
Phosphate at 0.01 M is one of the main buffers in blood plasma, whose pH is 7.45. Would phosphate be as useful if the plasma pH were 8.5?
-
Figure 8.5 illustrates the market for chocolate bars. If a new tax of $1.50 a chocolate bar is imposed, what is the change in the quantity of chocolate bars bought, who pays most of the tax, and what...
-
Suppose you bought a GM corporate bond on January 25, 2001 for $750, on January 25, 2004 sold it for $650.00. 6 What was your annual holding period return?
-
Coco bought a warehouse for $ 1 7 0 , 0 0 0 . depreciated it $ 1 8 , 0 0 0 , and sold it for $ 3 2 0 , 0 0 0 . What is the amount of the gain reported on the sale?
-
Cost of 1 2 % the project requires investment cost $ 4 8 0 0 today and is expected to straight free cash flow of $ 2 0 0 0 per year for the next five years. What is the project equivalent annual...
-
Two bonds A and B have the same credit rating, the same par value and the same coupon rate. Bond A has 30 years to maturity and bond B has five (5) years to maturity. Please demonstrate your...
-
On July 1, 2025, Coronado Inc. made two sales. 1. 2. It sold land having a fair value of $909,890 in exchange for a 4-year zero-interest-bearing promissory note in the face amount of $1,431,725. The...
-
What is the market share variance, and is it controllable by managers?
-
Assume Eq. 6-14 gives the drag force on a pilot plus ejection seat just after they are ejected from a plane traveling horizontally at 1300 km/h. Assume also that the mass of the seat is equal to the...
-
A 10.0-mL solution of 0.050 0 M AgNO 3 was titrated with 0.025 0 M NaBr in the cell S.C.E. 7 || titration solution | Ag(s) Find the cell voltage for 0.1 and 30.0 mL of titrant.
-
A solution containing 50.0 mL of 0.100 M EDTA buffered to pH 10.00 was titrated with 50.0 mL of 0.020 0 M Hg(ClO 4 ) 2 in the cell shown in Exercise 14-B: S.C.E. 7 titration solution | Hg(l) From the...
-
Consider the titration in Figure 15-2. (a) Write a balanced titration reaction. (b) Write two different half-reactions for the indicator electrode. (c) Write two different Nernst equations for the...
-
Required: 1. Use the indirect method to determine the net cash provided by operating activities for the year. 2. Prepare a statement of cash flows for the year.
-
Net Present Value Method, Present Value Index, and Analysis for a service company Continental Railroad Company is evaluating three capital investment proposals by using the net present value method....
-
Current Attempt in Progress Crane Corp. is considering purchasing one of two new diagnostic machines. Either machine would make it possible for the company to bid on jobs that it currently isn't...

Study smarter with the SolutionInn App


