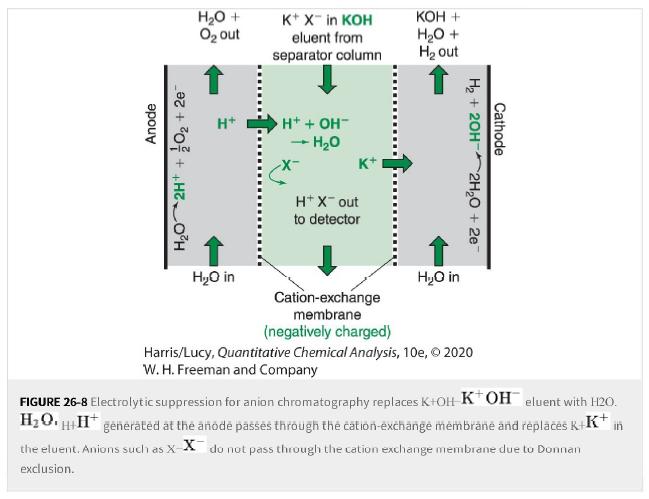
a. The suppressor in Figure 26-8 enables low parts per billion conductivity detection for anions such as
Question:
a. The suppressor in Figure 26-8 enables low parts per billion conductivity detection for anions such as Cl-Cl- and Br-,Br-. but very poor detection limits for anions such as CN-CN- and borate. Explain why.
b. Mixtures of sodium carbonate and bicarbonate can be used as eluent in suppressed-ion anion chromatography. Detection limits are poorer than with hydroxide eluent due to a higher background conductivity. Explain why.
Figure 26-8
Transcribed Image Text:
Anode H₂O 2H+ + O₂ +2e H₂O + O₂ out H+ H₂O in K+ X™ in KOH eluent from separator column H+ + OH™ + H₂O K+ H+ X-out to detector KOH + H₂O + H₂ out H₂O in H₂ + 2OH- Cathode 2H₂O + 2e Cation-exchange membrane (negatively charged) Harris/Lucy, Quantitative Chemical Analysis, 10e, © 2020 W. H. Freeman and Company FIGURE 26-8 Electrolytic suppression for anion chromatography replaces K+OH KOH eluent with H2O. H₂OH+H* generated at the anode passes through the cation-exchange membrane and replaces K+K+ in the eluent. Anions such as X-X do not pass through the cation exchange membrane due to Donnan exclusion.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (11 reviews)
Solution a The suppressor in Figure 268 enables low parts per billion conductiv...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
The carbonate ion (CO32-) can act as either a monodentate or a bidentate ligand. Draw a picture of CO32- coordinating to a metal ion as a bidentate and as a monodentate ligand. The carbonate ion can...
-
Explain why hydroxide ion catalyzes the reaction of piperidine with 2,4-dinitroanisole, but has no effect on the reaction of piperidine with 1-chloro-2,4-dinitrobenzene. piperidine
-
Why are some countries today much poorer than other countries? Are todays poor countries destined to always be poorer than todays rich countries? If so, explain why. If not, explain how todays poor...
-
Please show your work D To be a valid route, it must consist of a sequence of valid move. Each valid move is either going to the right for one block or going down for 1 block. T d) How many valid...
-
For the conditions of Exercise 12 and the data in Table 11.2, carry out a test of the following hypotheses: H0: 1 = 0, H1: 1 = 0.
-
Suppose the Schoof Company has this book value balance sheet: The notes payable are to banks, and the interest rate on this debt is 10%, the same as the rate on new bank loans. These bank loans are...
-
What are three economic factors that influence what people buy?
-
Paige Company has completed all of its operating budgets. The sales budget for the year shows 50,000 units and total sales of $2,000,000. The total unit cost of making one unit of sales is $22....
-
During late 2019, the voters of the City of Sniithville authorized tax-supported bond issues totaling $8,000,000 as partial financing for projects to construct the extension of streets and the...
-
A small private university is planning to start a volunteer football program. A random sample of alumni is surveyed. It was found that 250 were in favor of this program, 75 were opposed, and 25 had...
-
Conductivity and contactless conductivity detectors were developed for suppressed capillary ion chromatography. Observed peak heights in millivolts for bromide standards are in the table. a. Use...
-
Low iron concentration (as low as 0.02 nM 0.02 nM) in the open ocean limits phytoplankton growth. Preconcentration is required to determine such low concentrations. Trace Fe3+Fe 3+ from a large...
-
Put the data described in Exercise 2.4.25, FemaleTemp, into the Theory-Based Inference applet to answer (a)(b). a. Again, we want to test to see whether average female body temperature is different...
-
Information on four investment proposals is given below: Investment Proposal A B C Investment required Present value of cash inflows Net present value Life of the project $ (190,000) 267,400 $ 77,400...
-
What strategies can be employed to navigate the complexities of intercultural dynamics and foster inclusivity within globalized organizational frameworks ?
-
(e) Find principal value of sin-1 b a-2b (f) Find value of a, b, c, d from [2+ 4-2-11 24 15c d 4c+ 3d]
-
I need some direction with how to proceed with this. The project management plan would be for Customer Relationship Management (CRM) System for a Small Business. Thank you in advance PROJECT NAME...
-
(6) Solve the following logarithmic equations for x, show some work: (a) log4 + log4(x-3)= 1 (b) log(5-4x) = 2 log x (c) log+log(3x-5)= log 12 (d) log2 (7x+3)- log2 (2x-3)= 3
-
Watson Company has a subsidiary in the country of Alonza where the local currency unit is the kamel (KM). On December 31, 2012, the subsidiary has the following balance sheet: The subsidiary acquired...
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
State whether the errors in (a) - (d) are random or systematic: (a) A 25-mL transfer pipet consistently delivers 25.031 0.009 mL. (b) A 10 - mL buret consistently delivers 1.98 0.01 mL when drained...
-
State whether the errors in (a) - (d) are random or systematic: (a) A 25-mL transfer pipet consistently delivers 25.031 0.009 mL. (b) A 10 - mL buret consistently delivers 1.98 0.01 mL when drained...
-
Cheryl, Cynthia, Carmen, and Chastity shot the targets below at Girl Scout camp. Match each target with the proper description. (a) Accurate and precise (b) Accurate but not precise (c) Precise but...
-
If a company has total assets of $1632000; liabilities of $204000, common stock of $918000, and retained earnings of $510000 at December 31, 2025. What are the stockholders' claims on their assets at...
-
Select all that apply Under Section 4400, collections should be recorded: Multiple select question. at cost, consistently applied for all collections. nominal value, consistently applied for all...
-
The automatic GST exemption allocation for direct skip transfers applies as much of the transferor's remaining GST exemption as is necessary to create a zero inclusion ratio (or as close to zero as...

Study smarter with the SolutionInn App


