Calculate E and K for each of the following reactions. (a) I2(s) + 5Br2(aq) + 6H20 =
Question:
Calculate E and K for each of the following reactions.
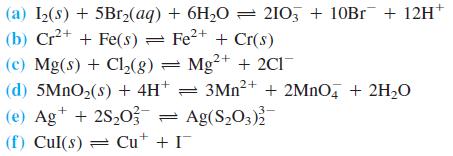
Transcribed Image Text:
(a) I2(s) + 5Br2(aq) + 6H20 = 210, + 10Br + 12H+ (b) Cr2+ + Fe(s) = Fe2+ + Cr(s) (c) Mg(s) + Cl,(g) = Mg²+ + 2C1 (d) 5MNO2(s) + 4H* = 3MN²+ + 2MNO, + 2H2O (e) Ag+ + 2S,03 = Ag(S,O3) (f) Cul(s) = Cu* + I
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
To calculate the standard potential E and equilibrium constant K for each of the given reactions we ...View the full answer

Answered By

Nithesh M
This is Vijaya Chandra, I was graduated from JNTU Engineering College in Mechanical Engineering. I have always shown a keen interest in physics, which is evident from his exceptional academic record in the subject. During my academic years, I was excelled in physics, consistently achieving high grades and performing well in exams. In addition to academic background, I worked as a tutor for 5 years in local village by helping students of all ages and skill levels achieve their goals. As a tutor, I have taught students of all classes for a period of five years, including graduate-level physics and have a deep understanding of the subject and passion for teaching has allowed me to develop unique teaching methods that make complex concepts easy to understand. I am comfortable working with students one-on-one or in small groups, and I am dedicated to helping them build confidence and succeed in their studies. Outside tutoring, I enjoyed playing cricket and surfing internet browser.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Predict the principal organic product of each of the following reactions:
-
Identify the principal organic products of each of the following reactions:
-
Predict the major organic product in each of the following reactions:
-
You are the president of a new, capital-rich firm and you must decide on your firm's targeting approach. Three large subgroups exist in your market that have never had their unique needs served. If...
-
In a taste test of two chocolate chip cookie recipes, 13 out of 18 subjects favored recipe A. Using the sign test, find the significance probability when H1 states that recipe A is preferable.
-
Match the transaction from the first column with the type of accounting adjustment from the second column. 1. Record depreciation for the month on buildings. 2. Record rental income for the month on...
-
You are talking to a friend who says that she never sets any financial goals and that her financial success or failure happens by luck.
-
Callie Peters is completing the audit of MakingNewFriends.com for the year ended December 31, 2013. Callie has been the audit manager on this engagement for the past three years. MakingNewFriends.com...
-
Of the following statements, which is false? A. The availability of the unlimited marital deduction merely postpones the potential estate tax due. B. Property that is not included in the decedent's...
-
The Northshore Bank is working to develop an efficient work schedule for full-time and part-time tellers. The schedule must provide for efficient operation of the bank including adequate customer...
-
In olden days, mercury cells with the following chemistry were used to power heart pacemakers: What is the cell voltage? If the power required to operate the pacemaker is 0.010 0 W, how many...
-
Calculate the voltage of each of the following cells. With the reasoning in Figure 13-8, state the direction of electron flow. (a) Fe(s) | FeBr 2 (0.010 M) || NaBr(0.050 M) | Br 2 (l) | Pt(s) (b)...
-
Which of the following describes an Access aggregate function? a. An aggregate function performs a calculation that provides new information for each record. b. An aggregate function determines all...
-
In manufacturing its products, Trevano Corp. adds all direct material at the beginning of the production process. The company's direct labor and overhead are considered to be continuously at the same...
-
How did the leadership in the Canadian National Railway help grow their internal business processes to allow them to become the most efficient and effective company in its field? Discuss using eps...
-
Logan Passport Services budgeted sales of 8,100 units of its Basic Visa Service (BVS). This was based on its estimated 5 percent share of this service in the relevant regional market. Actual sales of...
-
3. Body language (non-verbal communication) can convey messages to others the same as verbal communication can. Explain how you can use each of the following examples of body language when serving...
-
What is the selling price to the customer of Job No. 2468? Direct materials consumed Direct labor rate per hour Production overhead per direct labor hour Direct labor hours employed Admin. other...
-
To what extent do not-for-profit organizations have the ability to choose among the following: a. using depreciation, b. ignoring depreciation, c. maintaining building and equipment on the balance...
-
What tools are available to help shoppers compare prices, features, and values and check other shoppers opinions?
-
Nitric oxide (NO) is a cell-signaling agent in physiologic processes including vasodilation, inhibition of clotting, and inflammation. A sensitive chromatography-mass spectrometry method was...
-
Equation 22-33 contains terms (A, B, and C) describing three band-broadening mechanisms. (a) Which term is 0 for an open tubular column? Why? (b) Express the value of B in terms of measurable...
-
As the inside radius of an open tubular column is decreased, the maximum possible column efficiency increases and sample capacity decreases. For a thin stationary phase that equilibrates rapidly with...
-
Lynda Shaffer's article is a 'revisionist' work. Having read this work, define revisionism. Provide examples from "Southernization" to show how her work challenged your view (or the dominant view as...
-
The following table contains the number of successes and failures for three categories of a variable. Test whether the proportions are equal for each category at the a=0.01 level of significance....
-
9. x = log3 200. An intermediate step is A. log10 200 10910 3 B. log103 10910 200 C. log 10 3 200 D. 200 log 10 3 10. What is one of the three pairs of polar coordinates for the point (5, -60) with...

Study smarter with the SolutionInn App


