Calculate the theoretical masses of the sucralose species in Figure 22-14 at nominal masses of 395,395, 397,397,
Question:
Calculate the theoretical masses of the sucralose species in Figure 22-14 at nominal masses of 395,395, 397,397, 399,399 and 401.401. Find the difference in ppm between observed and calculated m/z.m/z.
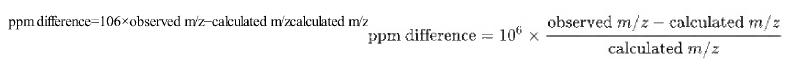
Figure 22-14

Transcribed Image Text:
ppm difference=106xobserved m/z-calculated m/zcalculated m/z ppm difference = 106 observed m/z - calculated m/z calculated m/z
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Answer Theoretical masses of the sucralose species in Figure 2214 at nominal masses of 39539...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
Calculate the theoretical masses of the species in Figure 21-9 and compare your answers with the values observed in the figure. Figure 21-9 2 000 31p+ 14N16OH* 1 000 15N160* 30.97 30.98 30.99 31.00...
-
In Figure 22-14, the sucralose species with a nominal mass X=395 X=395 is [12C121H1816O835C13]-. [ 12 C 12 1 H 18 16 O 8 35 Cl 3 ]-. X+1X + 1 arises from isotopologues containing one 13C, 13 C, one...
-
The point masses m and 2m lie along the x-axis, with m at the origin and 2m at x = L. A third point mass M is moved along the x-axis. (a) At what point is the net gravitational force on M due to the...
-
How to respond to the following? Cycle stock, pipeline inventory, buffer stock, and in-transit inventory all have associated carrying costs. Interest rates on inventory values, warehousing expenses,...
-
Let the statistic U2 be as defined by Eq. (11.3.32), and let be fixed positive constant. Show that for all observed values (xi , yi), for i = 1, . . . , n, the set of points (0, 1) such that U2 < ...
-
Explain the concept of structured programming. Why is it important?
-
On the Internet, locate the U.S. Dept. of Jus- tice, "Education and Training in Fraud and Forensic Accounting: A Guide for Education- al Institutions, Stakeholder Organizations, Faculty, and...
-
Juan Morales Company had the following account balances at year-end: Cost of Goods Sold $60,000; Inventory $15,000; Operating Expenses $29,000; Sales Revenue $115,000; Sales Discounts $1,200; and...
-
Number1. A machine (fixed asset) will be used starting January 1, 2011; acquisition cost of IDR 50,000,000; useful life 5 years; without residual value; depreciated using the straight-line method....
-
Multiple Choice Questions Select the best answer for each of the following situations and give reasons for your choice. a. Which of the following controls would most likely reduce the risk of...
-
Mass spectral interpretation. The compound C9H4N2Cl6 C 9 H 4 N 2 Cl 6 is a by-product found in chlorinated pesticides. a. Verify that the formula for rings + double bonds agrees with the structure....
-
When looking at the mass spectrum of an unknown substance, you tentatively identify the molecular ion MM as the peak with the most significant intensity at the high-mass end of the spectrum. Then you...
-
Officers, but not directors, owe a duty of loyalty to the corporation. (True/False)
-
1)Stefano has a perpetuity immediate that pays $1259.06 quarterly. Tne nominal interest rate is 2.010% compounded monthly. What is the present value of his perpetuity? 2)Keiko has a 18 year annuity...
-
1. Derive the equations x = [1 - e-kt] (cos a) g y = - + -[1 ekt](sin a) + 1/2 (1 kt ekt) - by solving the following initial value problem for a vector r in the plane Differential equation: dr dt =...
-
An EFT for $5 880 is received from Beck Ltd in payment of $6 000 for invoice number 109 dated 13 July. What was the discount rate on this payment?
-
In the figure above, the kinetic energy of a simple pendulum is plotted against its angle 0 from the vertical. If the mass of the pendulum bob is 1.28 kg, what is the length of the pendulum? Note...
-
Suppose that you are the project manager of a project with the following projed basellne information....estimated project duration of 4 months and an expected project cost of 400,000 How would you...
-
On December 1, 2013, Dresden Company (a U.S. company located in Albany, New York) purchases inventory from a foreign supplier for 60,000 local currency units (LCU). Dresden will pay in 90 days after...
-
Find the area of the surface generated by revolving the para- metric curve x = cos 1, y = sin? 1 (0 < I sa/2) about the y-axis.
-
Detection limit. A sensitive chromatographic method was developed to measure sub-part per-billion levels of the disinfectant by-products iodate (IO 3 ), chlorite (ClO 2 ), and bromate (BrO 3 ) in...
-
Olympic athletes are tested to see if they are using illegal performance-enhancing drugs. Suppose that urine samples are taken and analyzed and the rate of false positive results is 1%. Suppose also...
-
Olympic athletes are tested to see if they are using illegal performance-enhancing drugs. Suppose that urine samples are taken and analyzed and the rate of false positive results is 1%. Suppose also...
-
The following information is for Soweto Ltd: Product X: Revenue R15.00 Variable cost R2.50 Product Y: Revenue R25.00 Variable cost R10.00 Total fixed costs R50 000 If the sales mix shifts to one unit...
-
The home equity lending example discussed in class demonstrated.... Group of answer choices interest rate spread is the least important profitability factor waiving closing costs makes these products...
-
Prior service cost is recognized as pension expense Multiple choice question. over the future service period of the employees receiving the benefit. immediately when the retroactive amendment is...

Study smarter with the SolutionInn App


