Is it possible to precipitate 99.0% of 0.010 M Ce 3+ by adding oxalate (C 2 O
Question:
Is it possible to precipitate 99.0% of 0.010 M Ce3+ by adding oxalate (C2O42-) without precipitating 0.010 M Ca2+?
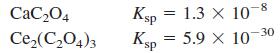
Transcribed Image Text:
CaC204 Ksp = 1.3 x 10-8 %3D Ce,(C,O4)3 Ksp 5.9 X 10-30
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Is it possible to make sound managerial decisions without business research? What advantages does research offer to the decision maker over seat-of-the-pants decision making?
-
Is it possible to design and enforce a law so that only honest citizens have access to guns?
-
Is it possible to use quantitative assessments of the organization's human resources to better link human resource management to firm strategy? Explain.
-
Selected accounts from the ledger of Restoration Arts for the fiscal year ended April 30, 2019, are as follows: Prepare a statement of owner's equity for the year. Apr 30 Doug Stone Capital Doug...
-
Mr. Z owns three homes. He lives in the San Francisco home full time. The other two he and his wife vacation in each year. He is considering replacing the house he has in Palm Springs, California,...
-
The Mann Corporation began operations in 2015. Information relating to the companys purchases of inventory and sales of products for 2015 and 2016 is presented below. Calculate the FIFO cost of goods...
-
LES is applied to a turbulent flow with convection heat transfer. Apply the filtering operation to (2.29) to derive the LES equation for temperature. Are there any terms that need modeling?
-
Multiple Choice. Choose the best answer. 1. Which of the following would not be considered a general long-term liability? a. The estimated liability to clean up the fuel and hazard waste storage...
-
. Sprint Fast Corporation, a CCPC, has the following in the current taxation year. . Taxable income: $790,000 . Income eligible for the Small Business Deduction: $490,000 . Aggregate investment...
-
The following balances come from the trial balance of Wilson State College as of the end of the 2020 fiscal year. Required a. Prepare a statement of revenues, expenses, and changes in net position...
-
Is it possible to precipitate 99.0% of 0.010 M Ce 3+ by adding oxalate (C 2 O 4 2- ) without precipitating 0.010 M Ca 2+ ?
-
For a solution of Ni 2+ and ethylenediamine, the following equilibrium constants apply at 20C: Calculate the concentration of free Ni 2+ in a solution prepared by mixing 0.100 mol of en plus 1.00 mL...
-
If the arguments for reducing income inequality and poverty are normative, why rely on the government to reduce the inequality? Why doesnt the private market resolve the problem?
-
Two rubber balls undergo an elastic collision. Ball 1 has a mass of 10 kg and an initial velocity of 5 m/s in the positive x direction. Ball 2 has a mass of 10 kg and is stationary. What is the final...
-
Calculate the angular momentum of a 265 kg motorcycle traveling at 25 m/s. traveling around a circular curve 500 m in radius
-
Use the spreadsheet template provided by your instructor to prepare the following operating budgets for the first quarter: (a) Sales budget/Cash collections Budget (b) Direct materials purchases...
-
Assume for a firm that budgeted production for July and August is 180,000 and 200,000 units, respectively. It takes half a kilogram of direct material to make one unit of finished product. Materials...
-
Shaver Co. manufactures a variety of electric razors for men and women. The company's plant is partially automated. Listed below is cost driver information used in the product-costing system:
-
Write a paper on global business legal issues with the United States working with Japan in the production of motor vehicles. What is the industry's supply chain? Who are its major players? What are...
-
In exchange for land, the company received a 12-month note on January 1. The face amount of the note is $1,000, and the stated rate of interest is 13%, compounded annually. The 13% rate is equal to...
-
Iminodiacetic acid forms 2:1 complexes with many metal ions: A 25.0 mL solution containing 0.120 M iminodiacetic acid buffered to pH 7.00 was titrated with 25.0 mL of 0.050 0 M Cu 2+ .Given that x2...
-
Iminodiacetic acid forms 2:1 complexes with many metal ions: A 25.0 mL solution containing 0.120 M iminodiacetic acid buffered to pH 7.00 was titrated with 25.0 mL of 0.050 0 M Cu 2+ .Given that x2...
-
What is the chelate effect?
-
The following companies have their financial year ended on 31 December 2021.1. Entity 1 has 1,000,000 ordinary shares at 1 each as on 1 January 2021. On 1 April 2021, the company issued 250,000 ord 2...
-
Coco Ltd. (Coco) purchased 90% of the voting shares of FerdinandInc. (Ferdinand) for $612,000 on January 1, 2022. On that date,Ferdinand's common shares and retained earnings were valued at$200,000 2...
-
. m 7) The program should be well-documented(comments) and easy to read. Bonus feature (Optional) 1) Allow the user to specify delivery ($5) or pickup ($10) and calculate the appropriate cost. The...

Study smarter with the SolutionInn App



