Table 19-1 gives examples of standard methods for analyses using a spectrophotometric reaction. a. What is a
Question:
Table 19-1 gives examples of standard methods for analyses using a spectrophotometric reaction.
a. What is a standard method of analysis?
b. U.S. Environmental Protection Agency method 350.1 for ammonia requires that a blank sample spiked with a known amount of analyte be analyzed within each batch of samples. What is the purpose of the spiked blank sample?
c. Standard Method 4500-Cl E for chloride states that particulate matter should be removed by filtration or centrifugation. Why must particulate be removed?
Table 19-1
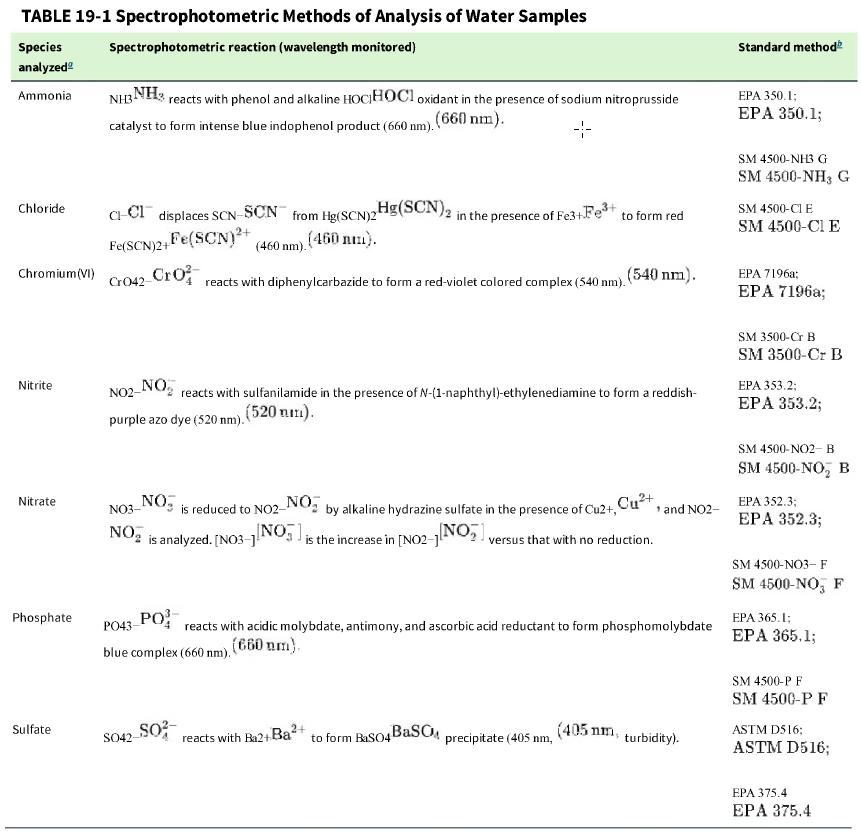
Transcribed Image Text:
TABLE 19-1 Spectrophotometric Species analyzed Ammonia Chloride Chromium (VI) Nitrite Nitrate Phosphate Sulfate Spectrophotometric reaction (wavelength monitored) NH3NH3 reacts with phenol and alkaline HOCHOCI oxidant in the presence of sodium nitroprusside catalyst to form intense blue indophenol product (660 nm). (660) nm). 4- Methods of Analysis of Water Samples C-Cl displaces SCN-SCN from Hg(SCN)2 Fe(SCN)2+ Fe(SCN)²+ (460 nm). (450 nm). Cro42-Cro NO2- NO purple azo dye (520 nm). reacts with diphenylcarbazide to form a red-violet colored complex (540 nm). ₂Hg(SCN) 2 in the presence of Fe3+Fe³+ to form red NO3- NO₂ is analyzed. [NO3-] SO42-SO² reacts with sulfanilamide in the presence of N-(1-naphthyl)-ethylenediamine to form a reddish- (520 mm). NO, NO₂ is reduced to NO2- by alkaline hydrazine sulfate in the presence of Cu2+, Cu²+, ¹ and NO2- [NO]; is the increase in [NO2-1[NO₂ versus that with no reduction. PO43- PO blue complex (660 nm). (660 nm). (540 nm). reacts with acidic molybdate, antimony, and ascorbic acid reductant to form phosphomolybdate 2+ reacts with Ba2+ Ba² to form BaSO4BaSOM precipitate (405 nm, (405 nm, turbidity). Standard method EPA 350.1; EPA 350.1; SM 4500-NH3 G SM 4500-NH3 G SM 4500-CI E SM 4500-CI E EPA 7196a; EPA 7196a; SM 3500-Cr B SM 3500-Cr B EPA 353.2; EPA 353.2; SM 4500-NO2-B SM 4500-NO, B EPA 352.3; EPA 352.3; SM 4500-NO3- F SM 4500-NO, F EPA 365.1; EPA 365.1; SM 4500-P F SM 4500-P F ASTM D516: ASTM D516; EPA 375.4 EPA 375.4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Answer a A standard method of analysis is a specific set of steps that must be followed ...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Question Posted:
Students also viewed these Sciences questions
-
The Environmental Protection Agency (EPA) requires that cities monitor over 80 contaminants in their drinking water. Samples from the Lake Huron Water Treatment Plant gave the results shown here....
-
A method used by the U.S. Environmental Protection Agency (EPA) for determining the concentration of ozone in air is to pass the air sample through a "bubbler" containing sodium iodide, which removes...
-
The Environmental Protection Agency took soil samples at 16 locations near a former industrial waste dump and checked each for evidence of toxic chemicals. They found no elevated levels of any...
-
8. A 12-tur square wire loop of 1-m length carrying a current of 1-A in the counter clockwise direction as shown in the figure. A magnetic field of magnitude 3000 G is directed into the board. Find,...
-
Suppose that 400 persons are chosen at random from a large population, and that each person in the sample specifies which one of five breakfast cereals she most prefers. For i = 1, . . . , 5, let pi...
-
Telemark-Kjemi AS manufactures an industrial solvent in two departments - mixing and cooking. This question focuses on the Cooking Department. During June 2007, 90 tones of solvent were completed and...
-
Determine whether the following are preventive (P), detective (D), or corrective (C) controls. a. Training b. Bank reconciliations c. Passwords d. Surprise cash count e. Quality circle team f. Job...
-
Haas Company is a retail company that specializes in selling outdoor camping equipment. The company is considering opening a new store on October 1, 2015. The company president formed a planning...
-
Please help. Compare the required credit risk capital under Basel | and Basel II for the following set of arrangements. (a) A 2 year interest rate swap with a principal of $100 million traded with an...
-
After taking business classes, Jake, an avid dog-lover, decided to start selling unique pet supplies at trade shows. He has two products: Product 1: "Launch-it"- a tennis ball thrower that will sell...
-
In the chapter opener, trace metals in teeth and bone provide information about Otzi the Icemans diet and environment as a child and adult. The inorganic mineral matrix of teeth and bone is...
-
Multivariate calibration. An important aspect of creating multivariate calibrations is selection of which and how many features (for example, wavelengths or wavenumbers) should be used to construct...
-
What is a search engine? Why are they important for todays businesses?
-
As you can guess, your project sponsor is very involved in this project. Now he is concerned about the risks associated with this website migration and what he believes could be a customer...
-
1. Why is the Cost of Retained Earnings the equivalent of the firm's own required rate of return on common stock? (1pt) 2. Calculate the after-tax cost of debt under each of the following conditions....
-
The Hat Store had the following series of transactions for Year 2 . Date Transaction Description Jan. 1 Beginning inventory 60 units@$22 Mar. 15 Purchased 2 2 0 units @ $ 2 6 . 0 0 May 30 Sold 1 5 0...
-
Consider the following very complex projects: The construction of a new elementary school in a busy urban center. List who you think would be your main stakeholders for the project you selected and...
-
A share of ABC Corp. is currently trading at $59 per share. European call and put options are available with an exercise price of $60 at a price of $4.74 and $3.51, respectively. They can be...
-
On January 1, 2012, Doone Corporation acquired 60 percent of the outstanding voting stock of Rockne Company for $300,000 consideration. At the acquisition date, the fair value of the 40 percent...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
Write the chemical reaction whose equilibrium constant is (a) Ka for benzoic acid, C6H5CO2H (b) Kb for benzoate ion, C6H5CO-2 (c) Kb for aniline, C6H5NH2 (d) Ka for anilinium ion, C6H5NH+3
-
Find the pH and fraction of dissociation () of a 0.100 M solution of the weak acid HA with Ka = 1.00 10-5.
-
Find the pH and fraction of dissociation () of a 0.100 M solution of the weak acid HA with Ka = 1.00 10-5.
-
Farmer Evans is holding 200 pounds of potatoes in storage for Chef Noble but Chef Noble has breached the contract by failing to pay for the potatoes. The potatoes are beginning to rot. If Farmer...
-
Average daily rate is calculated using: O Total Room Revenue and Total Number of Rooms Sold O Total Room Revenue. and Total Number of Rooms Available O Total Room Revenue and RevPAR
-
The more successfully a firm can align information technology with its business goals, the _ _ _ _ _ _ _ _ . Question content area bottom Part 1 A . more its data will cost B . less successful it...

Study smarter with the SolutionInn App


