When 22.63 mL of aqueous NaOH were added to 1.214 g of cyclohexylaminoethanesulfonic acid (FM 207.29, structure
Question:
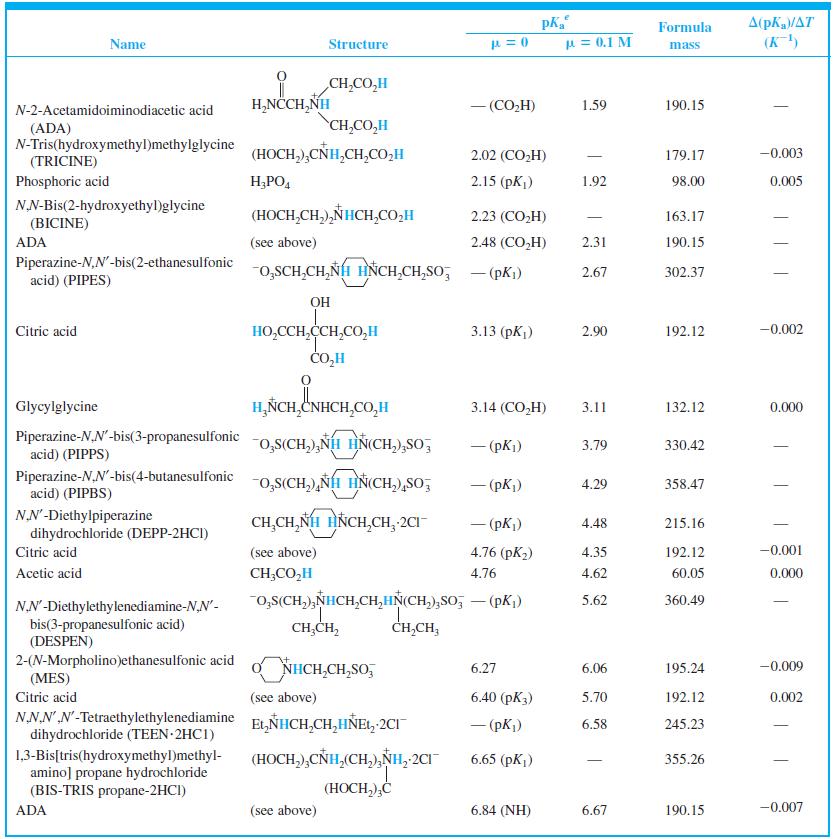
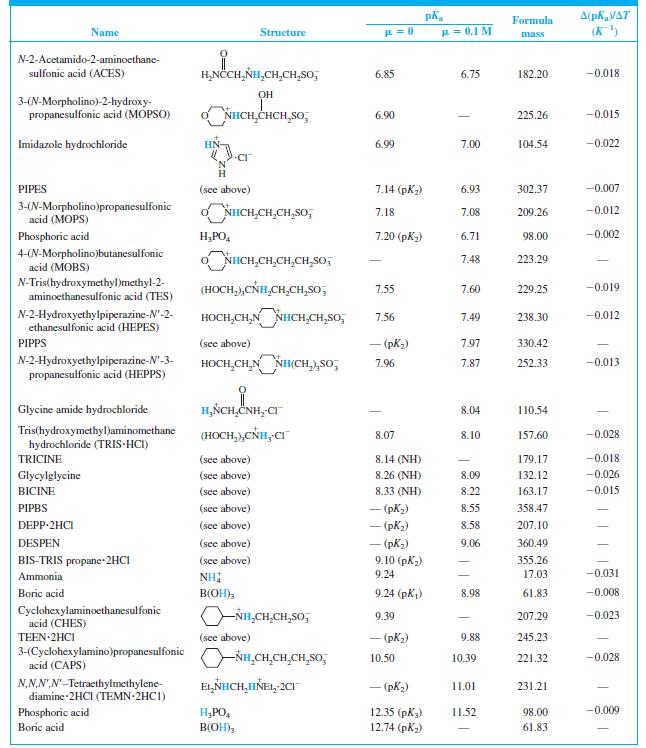
When 22.63 mL of aqueous NaOH were added to 1.214 g of cyclohexylaminoethanesulfonic acid (FM 207.29, structure in Table 8-2) dissolved in 41.37 mL of water, the pH was 9.24. Calculate the molarity of the NaOH.
Table 8-2
Transcribed Image Text:
A(pKa/AT (K) Formula Name Structure u = 0.1 M mass CH,CO,H H,NCCH,NH - (CO,H) 1.59 190.15 N-2-Acetamidoiminodiacetic acid CH,CO,H (ADA) N-Tris(hydroxymethyl)methylglycine (TRICINE) (HOCH,),CÑH,CH,CO,H 2.02 (CO,H) 179.17 -0.003 Phosphoric acid H;PO, 2.15 (pK) 1.92 98.00 0.005 NN-Bis(2-hydroxyethyl)glycine (HOCH,CH,),NHCH,CO,H 2.23 (CO,H) 163.17 | (BICINE) ADA (see above) 2.48 (СO,H) 2.31 190.15 Piperazine-N,N'-bis(2-ethanesulfonic acid) (PIPES) -o,SCH,CH,NH HNCH,CH,So; - (pKi) 2.67 302.37 OH HO,CCH,CCH,CO,H CO,H Citric acid 3.13 (pKj) 2.90 192.12 -0.002 Glycylglycine H,ÑCH,CNHCH,CO,H 3.14 (CO,H) 3.11 132.12 0.000 Piperazine-N,N'-bis(3-propanesulfonic acid) (PIPPS) "o,S(CH,),NÍH HN(CH,),SO, - (pK) 3.79 330.42 Piperazine-N,N'-bis(4-butanesulfonic acid) (PIPBS) "0,S(CH,),ÑH HN(CH,),SO, 4.29 358.47 ('yd) – NN'-Diethylpiperazine dihydrochloride (DEPP-2HCI) CH,CH, NH HNCH,CH, 2CI - (pK) 4.48 215.16 Citric acid (see above) 4.76 (pK2) 4.35 192.12 -0.001 Acetic acid CH;CO,H 4.76 4.62 60.05 0.000 o,s(CH,),NHCH,CH,HN(CH,),So, – (pK,) 5.62 360.49 NN'-Diethylethylenediamine-N,N'- bis(3-propanesulfonic acid) (DESPEN) CH,CH, CH,CH, 2-(N-Morpholino)ethanesulfonic acid (MES) Citric acid NHCH,CH,SO, 6.27 6.06 195.24 -0.009 (see above) 6.40 (рК3) 5.70 192.12 0.002 NN.NN'-Tetraethylethylenediamine dihydrochloride (TEEN 2HC1) EL,NHCH,CH,HÑE1, 2CI - (pK) 6.58 245.23 (HOCH,),CNH,(CH,),NH, 2CI 1,3-Bis[tris(hydroxymethyl)methyl- amino] propane hydrochloride (BIS-TRIS propane-2HCI) 6.65 (рK,) 355.26 (HOCH,),Ć ADA (see above) 6.84 (NH) 6.67 190.15 -0.007
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
So the reaction between acid and base is 11 mole ratio Volume of NaOH solution V 1 2263mL concent...View the full answer

Answered By

Parvathy D nair
Hi..it s me PARVATHY ..hails from KERALA ..i am a professional tutor and working in online platform.i have a experience of 2 years and solved more than 10000 answers through online platforms.i am a professional anchor also.i am a degree holder and doing bsc in zoology.currently waiting for MBBS admission.teaching is my passion.it s not only a job for me,,it s a devine profession.i see all my Students as my little brother and sisters..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
When 5.00 mL of 0.1032 M NaOH were added to 0.1123 g of alanine (FM 89.093) in 100.0 mL of 0.10 M KNO3, the measured pH was 9.57. Use activity coefficients to find pK2 for alanine. Consider the ionic...
-
When 100.0 mL of a weak acid were titrated with 0.093 81 M NaOH, 27.63 mL were required to reach the equivalence point. The pH at the equivalence point was 10.99. What was the pH when only 19.47 mL...
-
When an ?-hydroxy amide is treated with Br2 in aqueous NaOH under Hofmann rearrangement conditions, loss of CO2 occurs and a chain-shortened aldehyde is formed. Propose a mechanism. NH2 NH3 Br2...
-
Carlberg Company has two manufacturing departments, Assembly and Painting. The Assembly department started 11,000 units during November. The following production activity in both units and costs...
-
A taxpayer works for a law firm. You are working on her tax return. She tells you that she does not have any receipts for her daily parking because the firm pays the parking lot directly for an...
-
Helen Cole, her husband, and her son were partners in a business. Mrs. Cole's husband died, leaving his equity in the partnership to Mrs. Cole. Mrs. Cole and her son plan to form a new partnership...
-
On January 1, 2016, Pine Mountain Resorts acquires a 60 percent interest in Shawnee Peak Enterprises for \($25.4\) million in cash. The date-of-acquisition fair value of the noncontrolling interest...
-
IBS is a global provider of point-of-sale systems and related services that enable businesses to accept electronic payments. As a new hire in the companys international headquarters accounting...
-
rate is 5% per year? What is the total future value ten years from now of $400 received in 1 year, $350 received in 2 years and $900 received in 8 years if the interest O a. $2,047.15 b. $2, 129.89 O...
-
John Smith and Jane Brody are assistant portfolio managers. The senior portfolio manager has asked them to consider the acquisition of one of two option-free bond issues with the following...
-
Find the equilibrium constant for the reaction of MES (Table 8-2) with NaOH. Table 8-2 A(pKa/AT (K) Formula Name Structure u = 0.1 M mass CH,CO,H H,NCCH,NH - (CO,H) 1.59 190.15...
-
Why is the equivalence-point pH necessarily below 7 when a weak base is titrated with strong acid?
-
Suppose the concentration of a solute follows a Gaussian distribution (proportional to e x2 ) along the length of a container. Calculate the thermodynamic force on the solute at 20 C and 5.0 cm given...
-
First, read the case titled, "The Empowerment Plan (A): Providing Employment to Homeless Populations in Detroit." You will NOT be answering the questions listed at the end of case A. Reading this...
-
1. Reflect on the role of content communities and social networking sites, as social media, in terms of how the initial Greenpeace campaign escalated into a full-blown crisis for Nestle. 2. How would...
-
Satire is often classified as Horatian or Juvenalian (based on its similarity to the works of the Roman writers Horace and Juvenal). Horatian satire is relatively light and is meant to make us laugh...
-
What are some best practices when it comes to setting up a budget? What are the main areas to consider when creating a social media budget? What are some metrics you could develop for your specific...
-
Summarize the article shown below discussing the aspects of communication skills that university students have to be equipped with before they graduate from university. Based on your summarized...
-
1) Water is flowing at a rate of 6 m^3/min from a reservoir shaped like a hemispherical bowl of radius 13 m, shown here in profile. Answer the following questions, given that the volume of water in a...
-
Global.asax is used for: a. declare application variables O b. all other answers are wrong O c. declare global variables O d. handle application events
-
Scuba divers carry ballast weights to have neutral buoyancy. At that condition, the buoyancy force on the diver exactly balances weight, and there is no tendency either to float toward the surface or...
-
Examine the transition between the laminar and turbulent flows of water by sketching the stream of water that exits from a faucet (without an aerator) or hose (without a nozzle). You can control the...
-
Water flows through a 5-cm-diameter pipe at the average velocity of 1.25 m/s. (a) In the dimensions of L/s, what is the volumetric flow rate? (b) If the diameter of the pipe is reduced by 20% at a...
-
This paragraph explains the findings from research in the . From this research, what effects did leaded gasoline have on society? Check all that apply
-
5 . If a good is imported into ( large ) country H from country F , then the imposition of a tariff in country H A ) raises the price of the good in both countries ( the "Law of One Price" ) . B )...
-
Which one is true? Question 2 2 options: The demand curve is upward sloping. The supply curve is downward sloping. Expectations influence both buyers and sellers. Technical improvement shifts the...

Study smarter with the SolutionInn App


