1. If you dissolve 10.00 g of K3N (s) in 9.00 L of water (density =...
Fantastic news! We've Found the answer you've been seeking!
Question:
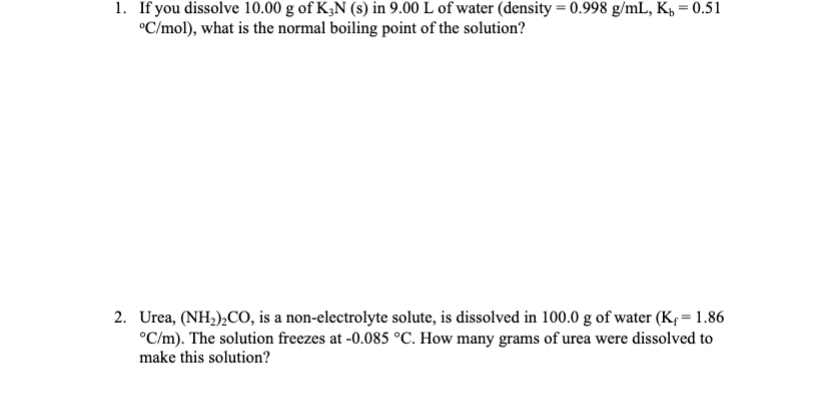
Transcribed Image Text:
1. If you dissolve 10.00 g of K3N (s) in 9.00 L of water (density = 0.998 g/mL, K = 0.51 C/mol), what is the normal boiling point of the solution? 2. Urea, (NH)CO, is a non-electrolyte solute, is dissolved in 100.0 g of water (K = 1.86 C/m). The solution freezes at -0.085 C. How many grams of urea were dissolved to make this solution? 1. If you dissolve 10.00 g of K3N (s) in 9.00 L of water (density = 0.998 g/mL, K = 0.51 C/mol), what is the normal boiling point of the solution? 2. Urea, (NH)CO, is a non-electrolyte solute, is dissolved in 100.0 g of water (K = 1.86 C/m). The solution freezes at -0.085 C. How many grams of urea were dissolved to make this solution?
Expert Answer:
Answer rating: 100% (QA)
Ill address each question separately 1 To find the normal boiling point of the solution for the compound KN we will use the formula for boiling point ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Identify relevant Commonwealth and state or territory WHS Acts, regulations, codes of practice, standards, guidance material and other relevant publications that will form the basis of a WHSMS
-
Create a cash flow sheet with Initial investment 100,000 resources expense : 5,000 labour cost: 10,000 maintenance cost : 3000 increase 2 warehouses (rental): 15,000 website development: 5,000...
-
You propose a new temperature scale with temperatures given in oM. You define 0.0oM to be the normal melting point of mercury and 100.0o to be the normal boiling point of mercury. (a) What is the...
-
Samson Company manufactures embroidered jackets. The company uses a standard cost system to control manufacturing costs. The following data represent the standard unit cost of a jacket: Fixed...
-
Describe the usual nature of the following features as they apply to a share of preferred stock: (a) Cumulative, and (b) Convertible.
-
I work on an assembly line. The first person creates the torso of a doll. I then get the torso's and I pop a head onto the doll. The next person down the line puts the arms and legs on the doll. And...
-
Use chloroform to extract acetone from water. Equilibrium data are given in Table 13-6. Find number of equilibrium stages required for a countercurrent cascade if feed is \(1000.0 \mathrm{~kg} /...
-
You work for Barbata Electronics. Your R&D people believe they have come up with an affordable technology that will double the capacity of existing MP3 players and uses audio format that is superior...
-
You ve started a part - time virtual bookkeeping business at the beginning of 2 0 2 3 . As of September 1 , 2 0 2 3 , you ve decided to incorporate your business, moved into a rented office space and...
-
Univex is a calendar year, accrual basis retail business. Its financial statements provide the following information for the year: Revenues from sales of goods $ 783,200 Cost of goods sold (FIFO) ...
-
The Elkmont Corporation needs to raise $52 million to finance its expansion into new markets. The company will sell new shares of equity via a general cash offering to raise the needed funds. The...
-
Write a Python function to perform a depth-first search (DFS) on a graph represented as an adjacency list
-
The Mermaid Hotel had 125 rooms sold at and ADR of $95.00 and the revenue potential is $14,500. What is the yield % for the Mermaid last night? (please show work below)
-
Julies' Merchandise has budgeted sales as follows for the third quarter of the year: July $ 4 4 , 0 0 0 August $ 7 5 , 0 0 0 September $ 6 4 , 0 0 0 Cost of goods sold in the company is equal to 7 0...
-
The fixed cost of B ltd. is $20,000, variable costs are $16,000 and break-even point is $8,000. You are required to find the sales and net profit of B ltd.
-
Project manager told his boss in a meeting that "Cost estimates and budgets are ideally based upon elements of the WBS and are prepared at the work package level. But when the cost cannot be...
-
Effort per hour, e 1 0.85 0.75 0.6 0 Reservation Status quo wage with higher unemployment Best response curve (with higher unemployment) reservation. wage Status quo best response curve Reservation...
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
Give the number of protons and electrons in each of the following common ions: K+, Mg2+, Fe3+, Br-, Mn2+, C4-, Cu2+.
-
`The standard enthalpies of formation of ions in aqueous solutions are obtained by arbitrarily assigning a value of zero to H+ ions; that is, Hf [H+(aq)] = 0. (a) For the following reaction ...
-
Methyl bromide (CH3Br, b.pt. 3.6C) is used as a soil fumigant to control insects and weeds. It is also a marine by-product. Photo dissociation of the C-Br bond produces Br atoms that can react with...
-
Metal Creations, Inc., is a custom manufacturer that uses a job order costing system. Currently, Metal Creations has 35% excess capacity in its factory. Charlie Rollins, the president, has instituted...
-
The CSR box in this chapter discusses CH2M Hills efforts to offer summer programs at no cost introducing underrepresented groups to STEM education. The companys stated goal is twofold: to increase...
-
For the graph below, write the adjacency matrix \(A\), compute \(A^{3}\), and verify that for each \(i\) and \(j, A^{3}(i, j)\) is the number of paths from \(i\) to \(j\) of length 3 by listing those...

Study smarter with the SolutionInn App


