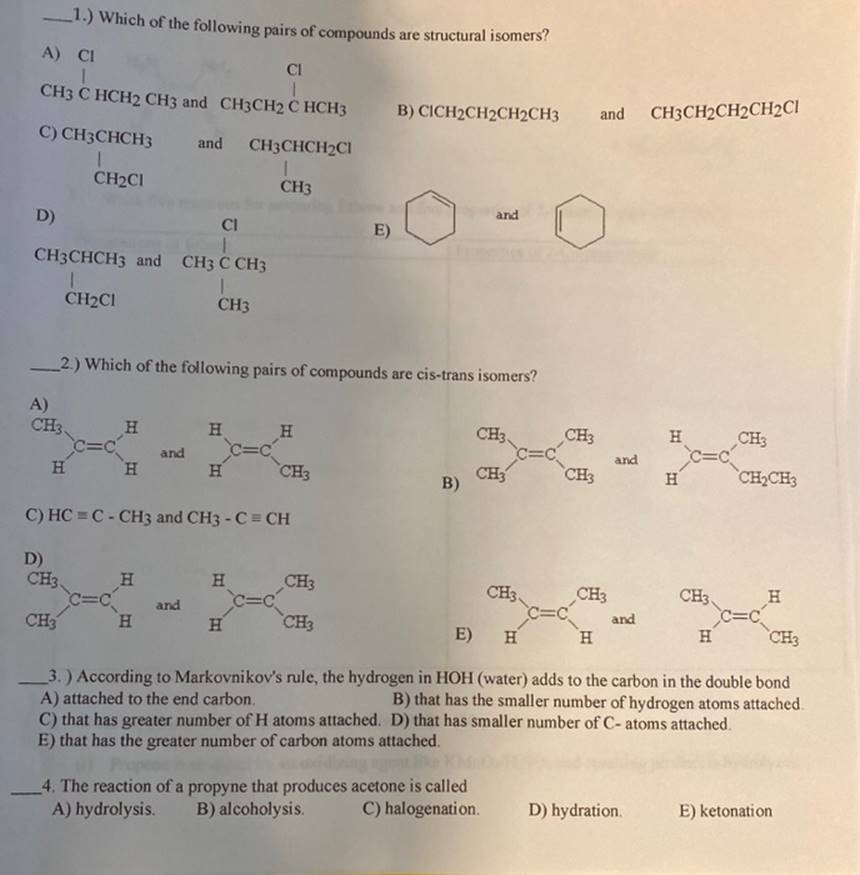
1.) Which of the following pairs of compounds are structural isomers? A) CI CI CH3 C...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1.) Which of the following pairs of compounds are structural isomers? A) CI CI CH3 C HCH2 CH3 and CH3CH2 C HCH3 B) CICH2CH2CH2CH3 and CH3CH2CH2CH2Cl C) CH3CHCH3 and CH3CHCH₂Cl 1 CH₂CI CH3 and D) CI E) CH3CHCH3 and CH3 C CH3 I I CH2C1 CH3 2.) Which of the following pairs of compounds are cis-trans isomers? A) CH3 H H H CH3, CH3 and H H H CH3 CH3 CH₂CH3 C) HC=C-CH3 and CH3 - C = CH D) CH3 H H CH3 CH3 CH3 H and C=C and CH3 H H CH3 E) H H H CH3 3.) According to Markovnikov's rule, the hydrogen in HOH (water) adds to the carbon in the double bond A) attached to the end carbon. B) that has the smaller number of hydrogen atoms attached. C) that has greater number of H atoms attached. D) that has smaller number of C- atoms attached. E) that has the greater number of carbon atoms attached. 4. The reaction of a propyne that produces acetone is called A) hydrolysis. B) alcoholysis. C) halogenation. D) hydration. E) ketonation C=C C=C C=C B) C=C CH3 CH3 and H C=C CH3 1.) Which of the following pairs of compounds are structural isomers? A) CI CI CH3 C HCH2 CH3 and CH3CH2 C HCH3 B) CICH2CH2CH2CH3 and CH3CH2CH2CH2Cl C) CH3CHCH3 and CH3CHCH₂Cl 1 CH₂CI CH3 and D) CI E) CH3CHCH3 and CH3 C CH3 I I CH2C1 CH3 2.) Which of the following pairs of compounds are cis-trans isomers? A) CH3 H H H CH3, CH3 and H H H CH3 CH3 CH₂CH3 C) HC=C-CH3 and CH3 - C = CH D) CH3 H H CH3 CH3 CH3 H and C=C and CH3 H H CH3 E) H H H CH3 3.) According to Markovnikov's rule, the hydrogen in HOH (water) adds to the carbon in the double bond A) attached to the end carbon. B) that has the smaller number of hydrogen atoms attached. C) that has greater number of H atoms attached. D) that has smaller number of C- atoms attached. E) that has the greater number of carbon atoms attached. 4. The reaction of a propyne that produces acetone is called A) hydrolysis. B) alcoholysis. C) halogenation. D) hydration. E) ketonation C=C C=C C=C B) C=C CH3 CH3 and H C=C CH3
Expert Answer:
Answer rating: 100% (QA)
Structural isomer can be defined as the compound having same chemical formula b... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Which of the following pairs of compounds could be separated by recrystallization or distillation? (a) meso-tartaric acid and (±)-tartaric acid (HOOC-CHOH-CHOH-COOH) (b) (c) (d) CH,CH, O CH,CH...
-
In each of the following pairs of compounds one is chiral and the other is achiral. Identify each compound as chiral or achiral, as appropriate. a. b. c. d. CICH CHCH OH and HOCH CHCH,OH CI OH CH,CH...
-
Which compound in each of the following pairs of compounds is the more soluble one? a. Silver chloride or silver iodide b. Magnesium hydroxide or copper(II) hydroxide
-
Ms. Esperanto obtained a $40,000 home equity loan at 7.5% compounded monthly. a. What will she pay monthly if the amortization period is 15 years? b. How much of the payment made at the end of the...
-
Multiple Choice Questions 1. Which of the following accounts would not appear in a closing entry? a. Interest Income b. Accumulated Depreciation c. Retained Earnings d. Salary Expense 2. Which...
-
Why do e-marketing plans need an evaluation component?
-
Identify responsibility centers (Learning Objective 5) Fill in the blanks with the phrase that best completes the sentence. A cost center A responsibility center Lower An investment center A profit...
-
The Eastern Oil Company buys crude vegetable oil. Refining this oil results in four products at the splitoff point: A, B, C, and D. Product C is fully processed by the splitoff point. Products A, B,...
-
Please answer question Question 5(Multiple Choice Worth 2 points) (04.02 MC) If an arithmetic sequence has terms as = 20 and 39 = 44, what is a15
-
On January 1, 20X5, Piper Ltd. purchased 100% of the shares of Sutton Ltd. for $ 1,085,000. At that time Sutton Ltd. had the following SFP: The bonds were issued at par and will mature in 10 years....
-
A vertical line drawn through a norma distribution at z=-0.60 separates the distribution into two sections, the body and the tail. Which proportion of the distribution is in the body?
-
You have completed the Learning Materials on Economics (Module 11) You have read this week's Course Reading: Bittman, M. (2014, Jul. 15). "The true cost of a burger." New York Times. Deliverables...
-
4. In November the US unemployment rate was 4.5% (USA Today, January 4, 2007). The Census Bureau includes nine states in the Northeast region. Suppose the random variable of interest is the number of...
-
Explore a case where employees feel demotivated due to limited career growth opportunities. How can HR create a career development framework that motivates employees to excel and grow within the...
-
What is the role of the financial manager in the flows of funds in the enterprise? 2. What is the main function of the financial markets in the financial environment? 3. Give 2 examples of financial...
-
SWOT analysis plays a role in strategic planning. Name the four components of SWOT analysis and explain in detail how each component is critical to strategic analysis. Identify which of the two forms...
-
10 Dikd rtgen eklindeki bir ka t zerine krokisi izilmi olan bir nehrin izledi i yolun yapt a lar ekildeki gibidir Nehrin izledi i yolun kar l kl kenarlan pa ralel oldu una g re m BCD a ka derecedir...
-
Show that the block upper triangular matrix A in Example 5 is invertible if and only if both A 11 and A 22 are invertible. Data from in Example 5 EXAMPLE 5 A matrix of the form A = [ A11 A12 0 A22 is...
-
Show how you would synthesize the following compounds, starting with benzene or toluene and any necessary acyclic reagents. Assume para is the major product (and separable from ortho) in ortho, para...
-
Use resonance forms to show delocalization of the negative charge in the Ruhemann's purple anion.
-
A student has just added ammonia to hexanoic acid and has begun to heat the mixture when he is called away to the telephone. After a long telephone conversation, he returns to find that the mixture...
-
Determine whether the sequence of edges represents an Euler circuit in Multigraph 15: \(K ightarrow L ightarrow N ightarrow M ightarrow O ightarrow S ightarrow T ightarrow Q ightarrow U ightarrow P...
-
The trial balance for Kilda Services, Inc., at the end of July appears at the top of the opposite page. It does not balance because of a number of errors. Kildas accountant compared the amounts in...
-
Give an example of a pair of edges that could be duplicated to eulerize Multigraph 14. Use the graphs and multigraphs in Figure 12.354.

Study smarter with the SolutionInn App


