6. The water-gas shift reaction (WGSR) is an important industrial reaction that is used in the...
Fantastic news! We've Found the answer you've been seeking!
Question:
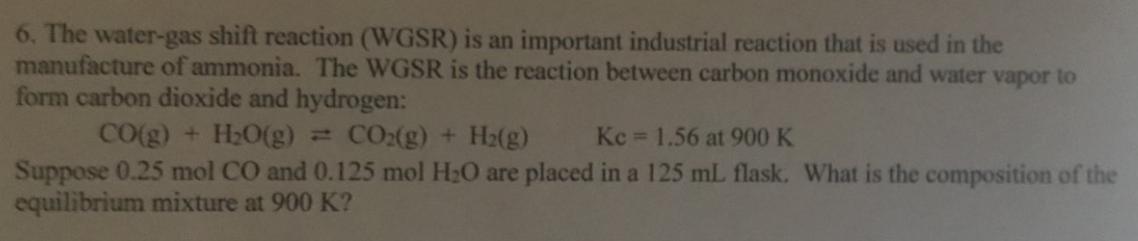
Transcribed Image Text:
6. The water-gas shift reaction (WGSR) is an important industrial reaction that is used in the manufacture of ammonia. The WGSR is the reaction between carbon monoxide and water vapor to form carbon dioxide and hydrogen: CO(g) + H20(g) = CO:(g) + H2(g) Kc 1.56 at 900 K Suppose 0.25 mol CO and 0.125 mol H20 are placed in a 125 mL flask. What is the composition of the equilibrium mixture at 900 K? 6. The water-gas shift reaction (WGSR) is an important industrial reaction that is used in the manufacture of ammonia. The WGSR is the reaction between carbon monoxide and water vapor to form carbon dioxide and hydrogen: CO(g) + H20(g) = CO:(g) + H2(g) Kc 1.56 at 900 K Suppose 0.25 mol CO and 0.125 mol H20 are placed in a 125 mL flask. What is the composition of the equilibrium mixture at 900 K?
Expert Answer:
Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these accounting questions
-
The strength of paper used in the manufacture of cardboard boxes (y) is related to the percentage of hardwood concentration in the original pulp (x). Under controlled conditions, a pilot plant...
-
A machine makes rivets that are used in the manufacture of airplanes. To be acceptable, the length of a rivet must be between 0.9 centimeter and 1.1 centimeters. The mean length of a rivet is 1.0...
-
A press produces parts used in the manufacture of large-screen plasma televisions. If the press is correctly adjusted, it produces parts with a scrap rate of 5%. If it is not adjusted correctly, it...
-
In the Edgeworth production box diagram, what conditions must hold for an allocation to be on the production contract curve? Why is a competitive equilibrium on the contract curve?
-
Under IAS 39, the IASB financial instrument standard in effect at the time, loans receivable were valued at amortized cost. That is, valuation was based on expected future receipts from the loan...
-
What is a hedge fund and how is it different from a mutual fund?
-
Use the statement of cash flows started in Application Problem 18-2. The comparative balance sheet of Flexcor Corporation lists the following long-term assets and their ending balances for the...
-
The Walmart retail chain sells standardized items and enjoys great purchasing clout with its suppliers, none of which it owns. The Limited retail chain sells fashion goods and owns Mast Industries,...
-
4-26 JOB COSTING, JOURNAL ENTRIES, T-ACCOUNTS, SOURCE DOCUMENTS. Production Company produces gadgets for the coveted small appliance market. The following data reflect activity for the most recent...
-
1) Use the base case assumptions as well as the information presented in the case to build a four-year discounted cash flow model for Advanced Seal given a 50% cannibalization rate for the Premium...
-
Elite Inc. has many divisions that are evaluated on the basis of return on investment (ROI). One division, Beta, makes boxes. A second division, Lambda, makes chocolates and needs 90,000 boxes per...
-
You need to either criticize or defend the philosopher's position. Do not do both. So, if you choose Kant, deductive argument that either criticizes or defends Kant's understanding of deontological...
-
4. Evaluate the following integrals: 4.1) arctan(x) dr 1+22 (Hint: tan(), tan(3)=3, tan() = 1 There should be no trig functions in your final answer.) 4.2) sin(x) In(cos(x)) cos(2) 4.3) 24-2 dr dr...
-
How do specific genotypes confer differential susceptibility or resistance to environmental stressors, and what are the evolutionary consequences of such genotype-dependent selection pressures ?
-
When we speak about project financing, which is the purspose of the Liquidity ratios? What do they determine and which are the main ones which should be considered?
-
Write about Working Capital Turnovers Ratio terms of Mirvac group $0.12 million, Boral ltd $4.54 million and lendlease corporation ltd $3413 million. go indepth about each number and how it will...
-
Question 1. (24 points) Context-free grammars (a) (10 points) Construct a context-free grammar that produces all (simplified) Prolog assertions with any finite number (1 or greater) of names. Such...
-
Three successive resonance frequencies in an organ pipe are 1310, 1834, and 2358 Hz. (a) Is the pipe closed at one end or open at both ends? (b) What is the fundamental frequency? (c) What is the...
-
A symmetric converging diverging duct with an area ratio of 2 is placed in wind tunnel where it encounters a 700 m/s flow of air at 50 kPa and 300 K. Determine the bypass ratio (diverted flow /...
-
Derive an expression for estimating the pressure (p) at which graphite and diamond exist in equilibrium at 300 K and 100 kPa in terms of the specific Gibbs function.
-
A refrigerant is compressed by an adiabatic compressor operating at steady state to raise the pressure from 200 kPa to 750 kPa. The following data are supplied for the inlet and exit ports. Inlet: v...
-
A reservoir at vertex 1 in the diagram below is to supply water to several pumping stations. The edge weights are costs of laying pipe from one station to another. How should the pipe be laid so that...
-
Identify which internal control concept is being violated and explain how this may cause an opportunity for fraud to occur within a business: 1. The supervisor for the purchasing department has not...
-
All of the following are typically included in the cash and cash equivalents account except: a. Cash b. Marketable securities c. Money market funds d. 30-day U.S. Treasury bills

Study smarter with the SolutionInn App



