A handbook lists the density of lead as 11.3 g/mL. Several groups of students are attempting...
Fantastic news! We've Found the answer you've been seeking!
Question:
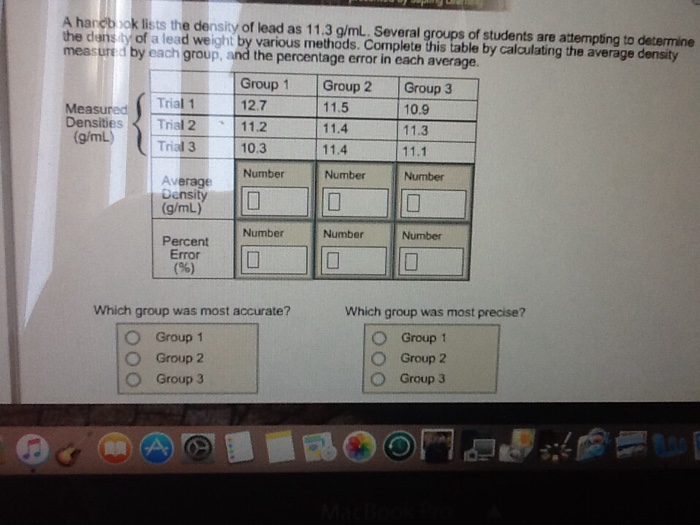
Transcribed Image Text:
A handbook lists the density of lead as 11.3 g/mL. Several groups of students are attempting to determine the density of a lead weight by various methods. Complete this table by calculating the average density measured by each group, and the percentage error in each average. Measured Densities (g/mL) Trial 1 Trial 2 Trial 3 Average Density (g/mL) Percent Error (%) A Group 1 12.7 11.2 10.3 Number Which group was most accurate? Group 1 Group 2 Group 3 **** Number 0 Group 2 11.5 11.4 11.4 Number Number 0 Group 3 10.9 11.3 11.1 Number 0 Number 0 Which group was most precise? Group 1 Group 2 Group 3 A handbook lists the density of lead as 11.3 g/mL. Several groups of students are attempting to determine the density of a lead weight by various methods. Complete this table by calculating the average density measured by each group, and the percentage error in each average. Measured Densities (g/mL) Trial 1 Trial 2 Trial 3 Average Density (g/mL) Percent Error (%) A Group 1 12.7 11.2 10.3 Number Which group was most accurate? Group 1 Group 2 Group 3 **** Number 0 Group 2 11.5 11.4 11.4 Number Number 0 Group 3 10.9 11.3 11.1 Number 0 Number 0 Which group was most precise? Group 1 Group 2 Group 3
Expert Answer:
Answer rating: 100% (QA)
Group 1 Calculate the average density gmL as follows Density Trial 1Trial 2 Trial 3 Avera... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The density of lead is 11.3 g/cm3. What is this value in kilograms per cubic meter?
-
The accountants for a city government are attempting to determine the appropriate method of reporting a series of unique financial transactions. They have reviewed all of the authoritative sources of...
-
The accountants for a city government are attempting to determine the appropriate method of reporting a series of unique financial transactions. They review all of the authoritative sources of United...
-
There is a bond on the spot-market. Price is 87.63 USD. Risk-free interest rate is 1.22%. The forward-price is 102.78 USD. Is there any arbitrage possibility if time to maturity is 9 months?
-
Ruth Swazey, divisional controller and certified management accountant, was upset by a recent memo she received from the divisional manager, Paul Chesser. Ruth was scheduled to present the divisions...
-
A chemical company produces amongst its product range two industrial cleaning fluids, A and B. These products are manufactured jointly. Total sales are expected to be restricted because home trade...
-
In order to determine how risky a particular company is that you are auditing, you prepare these five ratios along with the same ratios of this company's peers: What are your thoughts about the risk...
-
Consider the PortaCom project discussed in Section 16.1. a. An engineer on the product development team believes that first-year sales for the new printer will be 20,000 units. Using estimates of $45...
-
My answer isn't listed. Please help. ew Policies rrent Attempt in Progress On July 1, 2019, Vaughn Manufacturing issued 8% bonds in the face amount of $11000000, which mature on July 1, 2025. The...
-
Consolidation related simulation example: Millennium Capital Management, Inc., (MCM) acquired a 90% interest in NextGen, Inc. MCM's Financial Manager, Matthew Steven, has prepared a draft memo to the...
-
The parameters of all elements in the circuit shown below are given: R=10.8 kn, R2=27.5 kn, R3=1.4 kn, C=3.92 F, and -9.7 V (the internal resistance of the battery is negligible). ww R ww R3 S R w...
-
Take a look at the picture below. What happens when the Beast becomes the Beau and Beauty becomes the Beast? How does the story change? How do our stereotypes about Gender Roles impact our thought...
-
What is a situation when you were stereotyped or stereotyped someone? What happened, what was the stereotype, and how it affected communication in the situation? How the situation could have been...
-
The diagram below shows three charges fixed in the positions Q3 120 0.10 m - Q 0.15 m- Q1=-1.0*10^-6 C, Q2 = +3.0* 10^-6 C and Q3 = -2.0* 10^-6 C. Calculate the resultant force on charge Q 1....
-
What social and cultural purposes or functions do marriages provide? Think cross-culturally. What social and cultural purposes or functions do families provide? What social and cultural purposes or...
-
In 1987, Gordon and Amy Tan opened a drug store in the city of Newton. During the next 30 years, the Tans devoted themselves to developing their business and to raising their four children. In 2017,...
-
A cupcake store sells 1000 cupcake per shift. Every day the store open for 2 shifts . Each shift is 7 hours , to service the customers each shift has 20 employees. who are paid an hourly rate of $25....
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Based on Lewis structures, predict the ordering of N-O bond lengths in NO+, NO2- and NO3-.
-
Using only the periodic table as your guide, select the most electronegative atom in each of the following sets: (a) Na, Mg, K, Ca; (b) P, S, As, Se; (c) Be, B, C, Si; (d) Zn, Ge, Ga, As.
-
A system goes from state 1 to state 2 and back to state 1. (a) What is the relationship between the value of E for going from state 1 to state 2 to that for going from state 2 back to state 1? (b)...
-
What is your taxable income?
-
What is the relationship between taxes and personal financial planning?
-
As budget director for a small state, you must raise $350 million for emergency repairs to the states bridges. You have several ways to fund the project. Your options include: place a sales tax of 5...

Study smarter with the SolutionInn App


