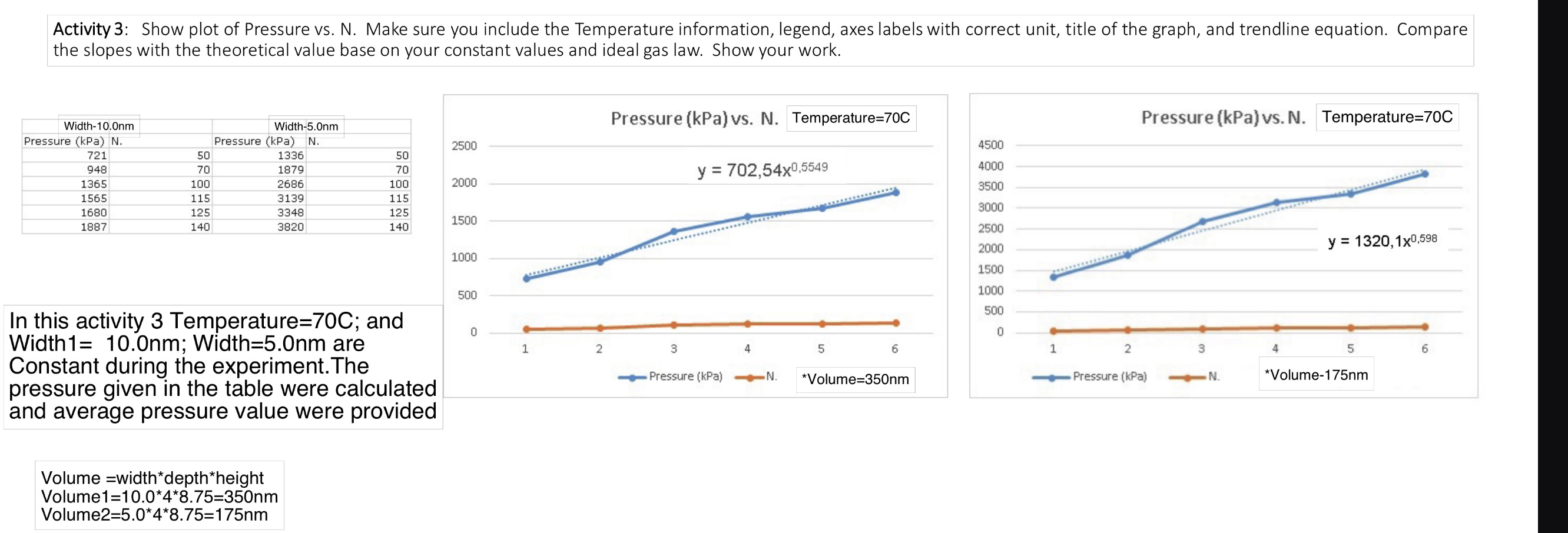
Activity 3: Show plot of Pressure vs. N. Make sure you include the Temperature information, legend,...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
Activity 3: Show plot of Pressure vs. N. Make sure you include the Temperature information, legend, axes labels with correct unit, title of the graph, and trendline equation. Compare the slopes with the theoretical value base on your constant values and ideal gas law. Show your work. Width-10.0nm Width-5.0nm Pressure (kPa) N. 721 Pressure (kPa) N. 2500 50 1336 50 948 70 1879 70 1365 100 2686 100 2000 1565 115 3139 115 1680 125 3348 125 1500 1887 140 3820 140 In this activity 3 Temperature=70C; and Width1= 10.0nm; Width=5.0nm are Constant during the experiment. The pressure given in the table were calculated and average pressure value were provided Volume width*depth*height Volume1=10.0*4*8.75=350nm Volume2=5.0*4*8.75=175nm Pressure (kPa) vs. N. Temperature=70C Pressure (kPa) vs. N. Temperature=70C 4500 y = 702,54x0,5549 4000 3500 3000 2500 2000 y=1320,1x0,598 1000 1500 1000 500 500 0 1 2 3 0 Pressure (kPa) 4 5 N. 6 1 2 3 4 5 6 *Volume=350nm Pressure (kPa) N. *Volume-175nm Physics 5B Lab 2 Remote Manual: Ideal Gas Laws UCLA Department of Physics and Astronomy In this lab, we will use the Gas Properties PhET Simulation to explore the relationships between pressure, volume, temperature, and particle number of gas. When an ideal system (with non-interacting particles) is closed, the total number of particles (N is held constant. Then, if either N, R, or T are held constant, then the other two physical parameter (p and V) are directly correlated, and there is either a proportional or inversely proportion relationship. This physics model of an ideal gas is very specific and only perfectly applicable ove a small range of physical situations. However, relationships between the physical parameters hol true in a qualitative sense for a much wider range of physical situations. Theory: Ideal gas law: pV = nRT = NkT Run the simulation Gas Properties (colorado.edu) 1. Select "Ideal" tab II Cool 203 K Pressure 886 kPa Hold Constant O Nothing Volume (V) Temperature (T) Pressure V Pressure T Width -- Stopwatch Collision Counter Particles Heavy Light 107 Gas Properties Ideal Explore Energy Diffusion PHET 2. Make sure you check the "Nothing" box and "Width" under tab (See the above figure). 3. Change the temperature reading to C. 4. The container depth (4 nm) and height (8.75 nm) are constant. You can change the volume by changing the width between 5 and 15 nm. Physics 5B Lab 2 Remote Manual: Ideal Gas Laws UCLA Department of Physics and Astronomy Activity 3: Constant T and V 1. Select either "Heavy" or "Light" particles. Do not mix. Put in some particles. 2. Choose a temperature for your measurement and fix at this temperature for all measurements. Record the temperature value and check the "Temperature (T)" box under tab. 3. Record the width you use. Calculate the volume of the container. 4. Choose 6 different N values. 5. Record the Pressure for each N. The pressure continues to change in value. Wait for a couple seconds then record the smallest and largest pressure value to obtain the average pressure value. 6. Plot Pressure vs. N. Include a trendline and trendline equation in the graph. 7. Repeat step 3 with a different volume value. Do not change the temperature. 8. Plot Pressure vs. N in the same graph in step 5. Include a trendline and trendline equation. 9. Make sure label the axes with correct unit and title of the graph (including the Temperature information). Include the legend showing the Volume of the curve. 10. Compare the slopes with the theoretical value base on your constant values and ideal gas law. Show your work. Activity 3: Show plot of Pressure vs. N. Make sure you include the Temperature information, legend, axes labels with correct unit, title of the graph, and trendline equation. Compare the slopes with the theoretical value base on your constant values and ideal gas law. Show your work. Width-10.0nm Width-5.0nm Pressure (kPa) N. 721 Pressure (kPa) N. 2500 50 1336 50 948 70 1879 70 1365 100 2686 100 2000 1565 115 3139 115 1680 125 3348 125 1500 1887 140 3820 140 In this activity 3 Temperature=70C; and Width1= 10.0nm; Width=5.0nm are Constant during the experiment. The pressure given in the table were calculated and average pressure value were provided Volume width*depth*height Volume1=10.0*4*8.75=350nm Volume2=5.0*4*8.75=175nm Pressure (kPa) vs. N. Temperature=70C Pressure (kPa) vs. N. Temperature=70C 4500 y = 702,54x0,5549 4000 3500 3000 2500 2000 y=1320,1x0,598 1000 1500 1000 500 500 0 1 2 3 0 Pressure (kPa) 4 5 N. 6 1 2 3 4 5 6 *Volume=350nm Pressure (kPa) N. *Volume-175nm Physics 5B Lab 2 Remote Manual: Ideal Gas Laws UCLA Department of Physics and Astronomy In this lab, we will use the Gas Properties PhET Simulation to explore the relationships between pressure, volume, temperature, and particle number of gas. When an ideal system (with non-interacting particles) is closed, the total number of particles (N is held constant. Then, if either N, R, or T are held constant, then the other two physical parameter (p and V) are directly correlated, and there is either a proportional or inversely proportion relationship. This physics model of an ideal gas is very specific and only perfectly applicable ove a small range of physical situations. However, relationships between the physical parameters hol true in a qualitative sense for a much wider range of physical situations. Theory: Ideal gas law: pV = nRT = NkT Run the simulation Gas Properties (colorado.edu) 1. Select "Ideal" tab II Cool 203 K Pressure 886 kPa Hold Constant O Nothing Volume (V) Temperature (T) Pressure V Pressure T Width -- Stopwatch Collision Counter Particles Heavy Light 107 Gas Properties Ideal Explore Energy Diffusion PHET 2. Make sure you check the "Nothing" box and "Width" under tab (See the above figure). 3. Change the temperature reading to C. 4. The container depth (4 nm) and height (8.75 nm) are constant. You can change the volume by changing the width between 5 and 15 nm. Physics 5B Lab 2 Remote Manual: Ideal Gas Laws UCLA Department of Physics and Astronomy Activity 3: Constant T and V 1. Select either "Heavy" or "Light" particles. Do not mix. Put in some particles. 2. Choose a temperature for your measurement and fix at this temperature for all measurements. Record the temperature value and check the "Temperature (T)" box under tab. 3. Record the width you use. Calculate the volume of the container. 4. Choose 6 different N values. 5. Record the Pressure for each N. The pressure continues to change in value. Wait for a couple seconds then record the smallest and largest pressure value to obtain the average pressure value. 6. Plot Pressure vs. N. Include a trendline and trendline equation in the graph. 7. Repeat step 3 with a different volume value. Do not change the temperature. 8. Plot Pressure vs. N in the same graph in step 5. Include a trendline and trendline equation. 9. Make sure label the axes with correct unit and title of the graph (including the Temperature information). Include the legend showing the Volume of the curve. 10. Compare the slopes with the theoretical value base on your constant values and ideal gas law. Show your work.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these physics questions
-
In a noninsurance situation, the exchange of unequal amounts sounds unfair. For example, Katie paid Nicolas $4,000 to repair her roof, but the repairs were actually only worth $1,000. Which one of...
-
Determine the tax liability for tax year 2022 in each of the following instances. In each case, assume the taxpayer can take only the standard deduction. Use the Tax Tables for taxpayers with taxable...
-
XYZ Inc. has a facility with six departments ( A - F ) . A summary of the processing sequence for 1 0 products and the weekly production forecasts for the products are given in the table below....
-
Using the DJIA data in Problem 5-39, use exponential smooth with trend adjustment to forecast the opening DJIA value for 2014. Use α = 0.8 and β = 0.2. Compare the MSE for...
-
Figure 16P-4 shows a local labor market for landscapers. What is the value of economic rent in this labor market?
-
An AM radio station broadcasts at 570 kHz. (a) What is the wavelength of the radio wave in air? (b) If a radio is tuned to this station and the inductance in the tuning circuit is 0.20 mH, what is...
-
P plc acquired 80% of the ordinary share capital of S plc for 150,000 and 50% of the issued 10% cumulative preference shares for 10,000, both purchases being effected on 1 May 2007. There have been...
-
What are the keys to Best Buys success? What are the risks going forward?
-
If tthe formula for the volume of a rectangular prism is V=Ixhxw where I = length, h = height and w = width. www Find the height of the figure below with the given volume. h 12 in 12 in V=1728 in
-
Kayviana is an active participant in the rental property that Kayviana owns. During the year, the property generates a ($19,000) loss; however, Kayviana has sufficient tax basis and at-risk amounts...
-
In exchange for a 40% interest in ABC Partnership, Faith contributes land with a basis of $50,000 and FMV of $125,000. There is a mortgage on the land for $30,000 which ABC Partnership assumes....
-
The diagram below shows a camera setup where an object is placed at a distance of 6m from a lens whose focal length is 8mm, where the diameter of the lens is 3cm f = 8mm Photosite d=3cm Image Sensor...
-
Find the roots of the system of equations below. Use an initial guess of x=y=4 and an error cutoff of 0.0001%. -x+xy+1.75 = 0 y+xy-x = 0 clear; clc; close all; Create a table of the iterations, x, y,...
-
At Company Y, the ratio of the number of female employees to the number of male employees is 3 to 2. If there are 150 female employees at the company, how many male employees are there at the company?
-
Analyze the narrative structure of metafictional works such as "House of Leaves" by Mark Z. Danielewski or "If on a winter's night a traveler" by Italo Calvino. How do these novels challenge...
-
Post 1 : Self-efficacy is an important concept in Bandura's work. If you believe in your capabilities to do something, and if you believe you have some control in a situation or the ability to affect...
-
It is possible to investigate the thermo chemical properties of hydrocarbons with molecular modeling methods. (a) Use electronic structure software to predict cHo values for the alkanes methane...
-
Jam Manufacturing Inc. has beginning work in process \($27,200\), direct materials used \($240,000\), di- rect labor \($200,000\), total manufacturing overhead \($150,000\), and ending work in...
-
Gene Toni claims that the distinction between directed indirect materials is based entirely on physical association with the product. Is Gene correct? Why?
-
Jane Diaz is confused about the differences between a product cost and a period cost. Explain the differ- ences to Jane.

Study smarter with the SolutionInn App


