All chemical reactions require that certain covalent bonds be broken within the reactants. In order for...
Fantastic news! We've Found the answer you've been seeking!
Question:
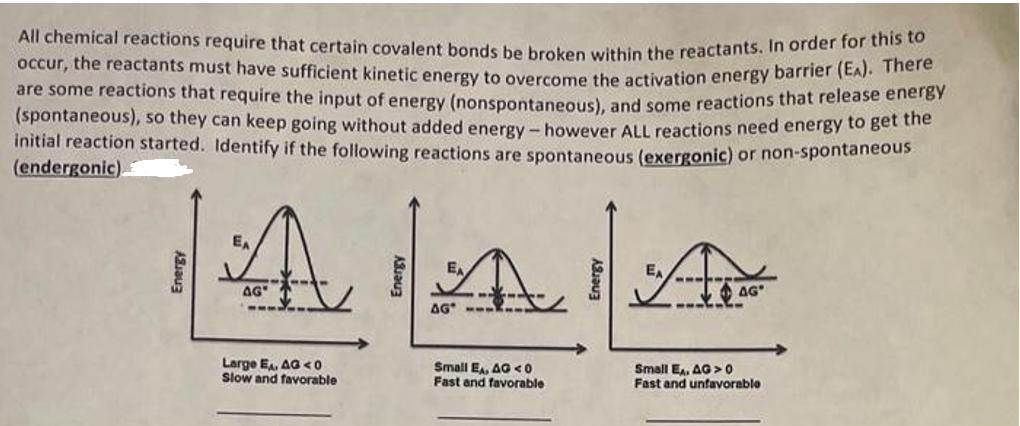
Transcribed Image Text:
All chemical reactions require that certain covalent bonds be broken within the reactants. In order for this to occur, the reactants must have sufficient kinetic energy to overcome the activation energy barrier (EA). There are some reactions that require the input of energy (nonspontaneous), and some reactions that release energy (spontaneous), so they can keep going without added energy - however ALL reactions need energy to get the initial reaction started. Identify if the following reactions are spontaneous (exergonic) or non-spontaneous (endergonic) Energy A AG" Energy ---, AA AG Large E, AG<0 Slow and favorable Small EA, AG40 Fast and favorable Small EA, AG>0 Fast and unfavorable All chemical reactions require that certain covalent bonds be broken within the reactants. In order for this to occur, the reactants must have sufficient kinetic energy to overcome the activation energy barrier (EA). There are some reactions that require the input of energy (nonspontaneous), and some reactions that release energy (spontaneous), so they can keep going without added energy - however ALL reactions need energy to get the initial reaction started. Identify if the following reactions are spontaneous (exergonic) or non-spontaneous (endergonic) Energy A AG" Energy ---, AA AG Large E, AG<0 Slow and favorable Small EA, AG40 Fast and favorable Small EA, AG>0 Fast and unfavorable
Expert Answer:
Answer rating: 100% (QA)
Solution Step1 1 The first reaction with large activation energy EA and negative G ie G 0 is spontan... View the full answer

Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these biology questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Suppose you come from a part of the world that is blessed with abundant water. Demand never comes close to the available amount. Should you be careful about the amount you use or should you simply...
-
What is the downward force exerted by the atmosphere on a soccer field whose dimensions are 105 m by 55 m?
-
Selected accounts from Lue Co.s adjusted trial balance for the year ended December 31 follow. Prepare a classified balance sheet. Total equity .. Equipment Salaries payable Accounts receivable Cash...
-
Find out what CFD software packages your college or university has. How many "seats" of each? How much does your institution spend annually on CFD software?
-
Business Objects trades on the Paris Bourse as ordinary shares and on the NASDAQ as American Depositary Receipts (ADRs). One ADR of Business Objects corresponds to one share on the Paris Bourse....
-
You have been hired as the new controller for the Ralston Company. Shortly after joining the company in 2024, you discover the following errors related to the 2022 and 2023 financial statements: a....
-
The Protein Chocolate Company 2019 sales are $30,046. Sales growth for 2020 is projected to grow 65% given new launches, increased spend and consumer interest. COGS was at 47% for 2019, and 42% for...
-
Coronado Company uses a job order cost system. On May 1, the company has a balance in Work in Process Inventory of $4,080 and two jobs in process: Job No. 429 $2,590, and Job No. 430 $1,490. During...
-
Define and contrast an Accounting Error with an Accounting Fraud. What differentiates them? Give an example.
-
Consider a reaction that consumes reactant C. Below are two plots of the same data set for two different functions containing the concentration of C in time, c(t) [c(t) has units of ng cm ]. Is the...
-
The numbers of raincoats sold in Classica department store over a three-year period were: Table 1 Quarter Year 1123 3 Summer 1 -166.875 Winter 248 272 304 Autumn 1 176.625 Winter 2 90.625 Spring 88...
-
Evaluating Corporate Social Responsibility - Corporate Social Responsibility 1 STARBUCKS https://www.starbucks.com/responsibility/global-report...
-
You are the human resources manager for an international company, and you will design a training presentation that you will use to train team leaders on being culturally aware of the diversity of the...
-
Express these numbers in standard notation. a. 2.87 10-8 b. 1.78 1011 c. 1.381 10-23
-
Use dimensional analysis to determine how the period T of a swinging pendulum (the elapsed time for a complete cycle of motion) depends on some, or all, of these properties: the length L of the...
-
A compass is placed directly on top of a wire (needle not shown). The current in the wire flows to the right. Which way does the north end of the needle point? Explain. (Ignore Earth's magnetic...
-
A long straight wire carries a 4.70-A current in the positive x -direction. At a particular instant, an electron moving at 1.00 Ã 107 m/s in the positive y -direction is 0.120 m from the wire....
-
Give some examples of how CRM-enabled sales analytics can provide valuable insights at each of these stages of the customer journey: prospect (lead), initial customer contact, engagement with the...
-
Someone says: Our firm focuses on maintaining long-term relationships with our customers. We dont have to do any prospecting. Evaluate this statement.
-
Why do some salespeople not immediately embrace the use of the newest technologies? What are some ways in which a salesperson who is reluctant to adopt new technologies might be influenced to do so?

Study smarter with the SolutionInn App


