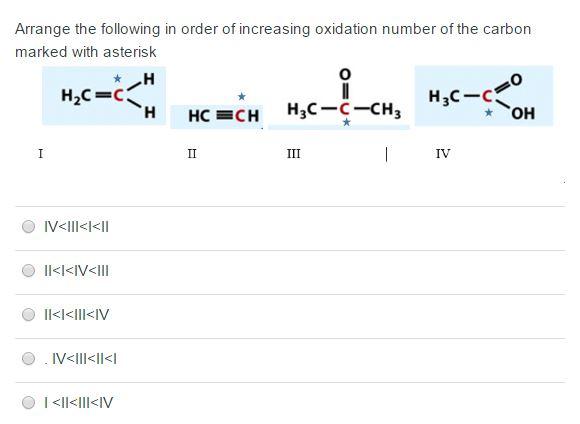
Arrange the following in order of increasing oxidation number of the carbon marked with asterisk *H...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Arrange the following in order of increasing oxidation number of the carbon marked with asterisk *H H I H₂C=C IV<III<I<II II<I<IV<III II<<|||<IV IV<III<II<I | <||<|||<IV HCECH II H3C-C-CH3 III 1 H3C-C - IV OH Arrange the following in order of increasing oxidation number of the carbon marked with asterisk *H H I H₂C=C IV<III<I<II II<I<IV<III II<<|||<IV IV<III<II<I | <||<|||<IV HCECH II H3C-C-CH3 III 1 H3C-C - IV OH
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Arrange the following species in order of increasing oxidation number of the sulfur atom: (a) H2S, (b) S8 (c) H2SO4, (d) S2-, (e) HS-, (f) SO2, (g) SO3.
-
Arrange the following in order of increasing radius and increasing ionization energy. a. N+, N, N- b. Se, Se-, Cl, Cl+ c. Br-, Rb+, Sr2+
-
Arrange the following in order of increasing ionic radius: Cl, Na+, and S2. Explain this order. (You may use a periodic table.)
-
ARTICLE: Reference Resolve Asset Management's Yes, You Can Eat Sharpe Ratios. 1- The author spoke to two ways for the foundation to meet its target return of 5%. Identify what the two ways presented...
-
The following values were obtained from the inventory records of the Harris Company, which has a fiscal year ending on December 31: Inventory, January 1, 2007, LIFO ..... $80,000 Inventory, March 31,...
-
The amount of DNA in an experiment increases rapidly from a very small value and then levels out at a large value before declining rapidly to 0. Draw graphs based on the above descriptions.
-
Sebel Corporation had cash flows from operating activities during the past year of $194,000. During the year, the company expended $25,000 for dividends; expended $158,000 for property, plant, and...
-
The stockholders equity section of Plaka Corporations balance sheet follows. Assuming one years dividend in arrears, determine the book value per share for both the preferred and the common stock....
-
Points: 0 of 1 A special purpose index has increased 109% during the last ten years. If the index is now 217, what was the index ten years ago? The index was (Round to the nearest integer as needed.)
-
Bruce also requires a calculation of taxable income for the most recent year ended December 31, 2019. The calculation should be prepared in Excel. He has also requested that you prepare the year-end...
-
2. How many latches will be generated after synthesis? Provide a brief explanation for your answer. library ieee; use ieee.std_logic_1164.all; entity test is port (a,b,use b: in STD_LOGIC; d out: out...
-
Two Na + ions are 3 . 6 nm apart. At 2 0 C what is the magnitude of the electric force between the ions if they are in seawater in which the positive and negative ions each have a concentration of 5...
-
Philippines Peso (the currency of Philippines) 1) Provide a plan of action that could be implemented to mitigate the impact of the currency movements, i.e., how you should to some firm approach...
-
Explain the investing and financing activities classifications on a statement of cash flows. Give your own example of each type of activity.
-
a) Explain three inventory valuation methods b) Discuss the implications of choosing different inventory methods on financial statement. c) How are these methods used to manipulate net income without...
-
the different types of budgets that are prepared along with their objectives and importance of those budgets in that particular industry. 1. Automobile Industry;
-
Countries with a strong individualism culture, (e.g., the United States, the UK, and Canada) are likely to have compensation systems with which of the following characteristics? Nonperformance variabl
-
Proposals have been made to ?sail? spacecraft to the outer solar system using the pressure of sunlight, or even to propel interstellar spacecraft with high-powered, Earth-based lasers. Sailing...
-
Calculate KP for the following reaction at 25C: H2(g) + I2(g) 2HI(g) G = 2.60 kJ/mol
-
Calculate the mass in grams of hydrogen chloride produced when 5.6 L of molecular hydrogen measured at STP react with an excess of molecular chlorine gas.
-
A 15.00-mL solution of potassium nitrate (KNO3) was diluted to 125.0 mL, and 25.00 mL of this solution were then diluted to 1.000 x 103 mL. The concentration of the nal solution is 0.00383 M....
-
Prepare summary performance report (Learning Objective 1) Hanna White owns a chain of travel goods stores. Last year, her sales staff sold 10,000 suitcases at an average sales price of $150. Variable...
-
Corporate headquarters expenses (Learning Objective 5) In Exhibit 10-20, the next to last line of the CEOs report consists entirely of expenses. Describe the kinds of expenses that would be included...
-
Management by exception (Learning Objective 5) Look at the performance report in Exhibit 10-20. According to the management by exception principle, on which variances should the manager of the...

Study smarter with the SolutionInn App



