5. As shown in the figure, a well-insulated turbine operating at steady state has two stages...
Fantastic news! We've Found the answer you've been seeking!
Question:
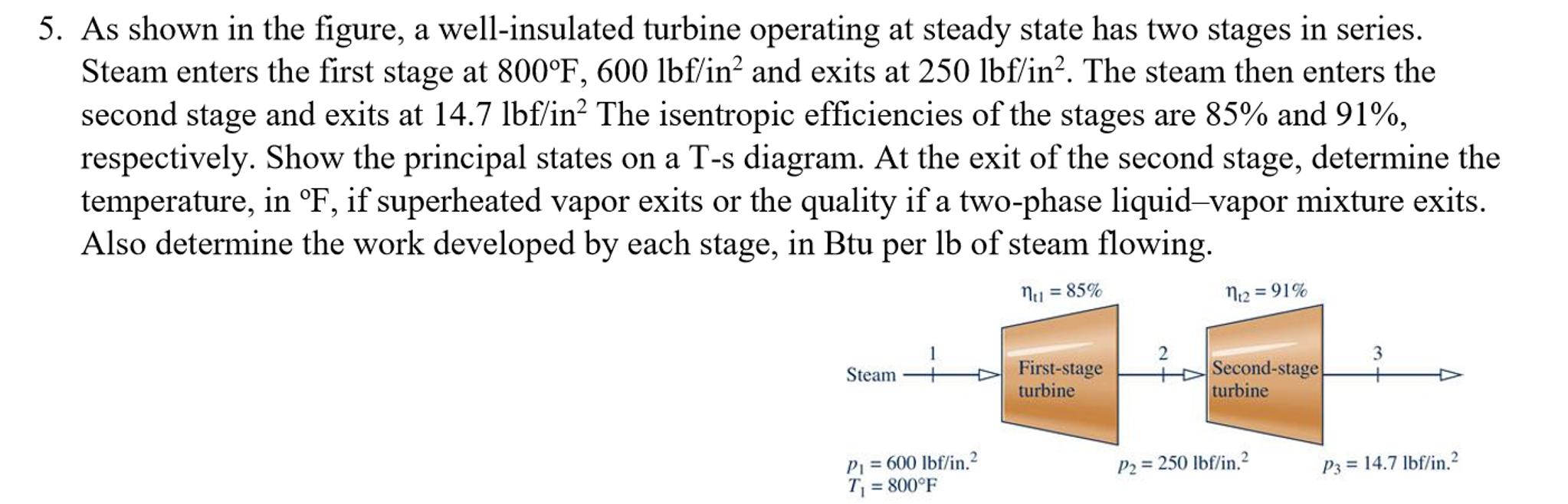
Transcribed Image Text:
5. As shown in the figure, a well-insulated turbine operating at steady state has two stages in series. Steam enters the first stage at 800°F, 600 lbf/in? and exits at 250 lbf/in?. The steam then enters the second stage and exits at 14.7 lbf/in? The isentropic efficiencies of the stages are 85% and 91%, respectively. Show the principal states on a T-s diagram. At the exit of the second stage, determine the temperature, in °F, if superheated vapor exits or the quality if a two-phase liquid-vapor mixture exits. Also determine the work developed by each stage, in Btu per lb of steam flowing. Nu = 85% N12 = 91% 3 First-stage turbine Second-stage turbine Steam P1 = 600 lbf/in.2 T = 800°F P2 = 250 lbf/in.? P3 = 14.7 lbf/in.? 5. As shown in the figure, a well-insulated turbine operating at steady state has two stages in series. Steam enters the first stage at 800°F, 600 lbf/in? and exits at 250 lbf/in?. The steam then enters the second stage and exits at 14.7 lbf/in? The isentropic efficiencies of the stages are 85% and 91%, respectively. Show the principal states on a T-s diagram. At the exit of the second stage, determine the temperature, in °F, if superheated vapor exits or the quality if a two-phase liquid-vapor mixture exits. Also determine the work developed by each stage, in Btu per lb of steam flowing. Nu = 85% N12 = 91% 3 First-stage turbine Second-stage turbine Steam P1 = 600 lbf/in.2 T = 800°F P2 = 250 lbf/in.? P3 = 14.7 lbf/in.?
Expert Answer:

Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these mechanical engineering questions
-
Steam enters a nozzle operating at steady state at 5 MPa and 420oC with negligible velocity, and exits at 3 MPa and 466 m/s. If the mass flow rate is 3 kg/s. Determine (a) The exit area, (b) The exit...
-
Two tanks are connected together as shown in Fig. P3.49, both containing water. Tank A is at 30 lbf/in.2, v = 8 ft3/lbm, V = 40 ft3 and tank B contains 8 lbm at 80 lbf/in. 2, 750 F. The valve is now...
-
A gas turbine operating at steady-state is shown in the following figure. Air enters the compressor with a mass flow rate of 5 kg/s at 0.95 bar and 22C and exits at 5.7 bar. The air then passes...
-
For data Tables in an Oracle Server database, a blank field must be a. Indicated by NULL Avoided b. Kept as a blank c. Indicated either by NULL or a blank d. Indicated by NULL
-
The enthalpy of neutralization for the reaction of a strong acid with a strong base is 256 kJ/ mol of water produced. How much energy will be released when 200.0 mL of 0.400 M HNO3 is mixed with...
-
A small rocket motor, fueled with hydrogen and oxygen, is tested on a thrust stand at a simulated altitude of \(10 \mathrm{~km}\). The motor is operated at chamber stagnation conditions of \(1500...
-
In terms of the importance of leadership, it has been argued that ____________. (a) leadership makes little or no difference. (b) only charismatic leadership is important. (c) charismatic leadership...
-
Herman and Sons Law Offices opened on January 1, 2015. During the first year of business, the company had the following transactions: January 2: The owners invested $ 250,000 (the par value of the...
-
PROBLEM 2 12 POINTS 25 MINUTES Board Games Ltd. provides you with the following information about its operations for the month of January 20X0. January 1 The company had 5,000 units of an item that...
-
Explain how you would determine how much profit was generated from orders placed in April 2009. Figure 1-5: ORDERS Order# Customer# Orderdate Shipdate Shipstreet Shipcity Shipstate Shipzip Shipcost...
-
Consider the following BNF grammar. S= A x | By A= By | CwBx |B w C y Which of the following regular expressions describes the same set of strings as the grammar? xwxy + xwwy +ywx xwx + xww*y + yw...
-
What are the major legal forms of intellectual property?
-
Discuss the suitability of bank loan financing for an entrepreneurial venture.
-
You are engaged in the audit of the financial statements of Armada Corporation for the year ended August \(31,200 \mathrm{X}\). The balance sheet, reflecting all of your audit adjustments accepted by...
-
How does one decide upon a valuation range? Why is the valuation range important?
-
Valley Corporation has a stock option plan designed to provide extra incentive to its officers and key employees. A note to the financial statements includes a description of the plan and lists the...
-
Tabitha, of Tabitha's Tasty Treats for Dogs, ran a regression for bags of organic treats sold and the related costs appears below. What is the total cost (rounded to the nearest whole dollar) if...
-
The production budget of Artest Company calls for 80,000 units to be produced. If it takes 30 minutes to make one unit and the direct labor rate is $16 per hour, what is the total budgeted direct...
-
Determine: (a) The work transfer involved in compressing a spring with a spring constant of 150 kN/m from its rest position by 10 cm. (b) What is the work done in compressing it further by another 10...
-
Air leaves a compressor in a pipe with a total temperature and pressure of 180oC, 350 kPa and a velocity of 150 m/s. The pipe has a cross sectional area of 0.02 m2. Determine (a) the static...
-
A solar-energy collector produces a maximum temperature of 100oC. The collected energy is used in a cyclic heat engine that operates in a 5oC environment. (a) What is the maximum thermal efficiency?...
-
Explain the concept of interest rate parity.
-
Jim Logan, owner of the Sports Exports Company, is concerned about the value of the British pound over time because his firm receives pounds as payment for footballs exported to the United Kingdom....
-
Recall that Blades, the U.S. manufacturer of roller blades, generates most of its revenue and incurs most of its expenses in the United States. However, the company has recently begun exporting...

Study smarter with the SolutionInn App


