a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest...
Fantastic news! We've Found the answer you've been seeking!
Question:
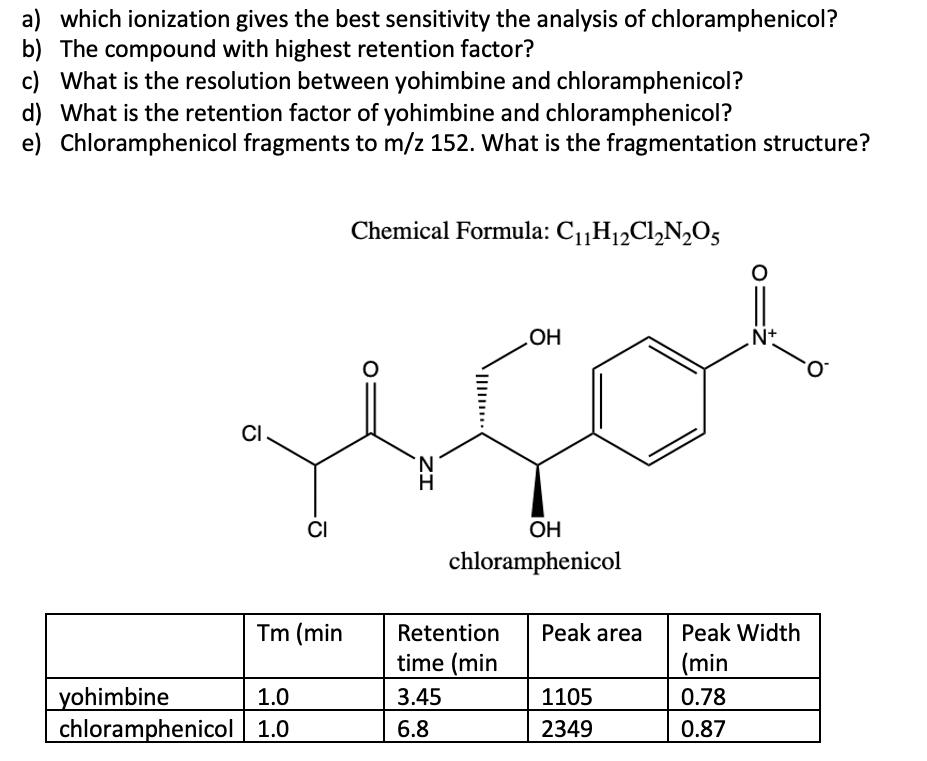
Transcribed Image Text:
a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What is the retention factor of yohimbine and chloramphenicol? e) Chloramphenicol fragments to m/z 152. What is the fragmentation structure? Chemical Formula: C1„H12C1,N,O5 HO Nt O. CI. CI OH chloramphenicol Tm (min Retention Peak area Peak Width time (min (min yohimbine chloramphenicol 1.0 1.0 3.45 1105 0.78 6.8 2349 0.87 ZI a) which ionization gives the best sensitivity the analysis of chloramphenicol? b) The compound with highest retention factor? c) What is the resolution between yohimbine and chloramphenicol? d) What is the retention factor of yohimbine and chloramphenicol? e) Chloramphenicol fragments to m/z 152. What is the fragmentation structure? Chemical Formula: C1„H12C1,N,O5 HO Nt O. CI. CI OH chloramphenicol Tm (min Retention Peak area Peak Width time (min (min yohimbine chloramphenicol 1.0 1.0 3.45 1105 0.78 6.8 2349 0.87 ZI
Expert Answer:
Answer rating: 100% (QA)
a The electrospray negative ionization Electrospray Ionization ESI probe in negative ionization mode ... View the full answer

Related Book For 

Applied Statistics in Business and Economics
ISBN: 978-0073521480
4th edition
Authors: David Doane, Lori Seward
Posted Date:
Students also viewed these chemistry questions
-
Compound C (C9H11NO) gives a positive Tollens' test (can be oxidized to a carboxylic acid) and is soluble in dilute HCl. The IR spectrum of C shows a strong band near 1695 cm-1 but shows no bands in...
-
A compound with molecular formula C4H6O gives the infrared spectrum shown in Figure 13.34. Identify the compound. 3.5 4 5 12 13
-
compound with molecular formula C5H10O2 gives the following IR spectrum. When it undergoes acid-catalyzed hydrolysis, the compound with the following 1H NMR spectrum is formed. Identify the...
-
Prove the following statement: If a transformer having a series impedance Ze is connected as an autotransformer, its per-unit series impedance Z as an autotransformer will be SE Zoq NSE + NC Note...
-
Gramercy, Inc., manufactures sailboats and has two major categories of overhead: materials handling and quality inspection. The costs expected for these categories for the coming year are as follows:...
-
How can the Taylor rule be used as a guide to evaluating Federal Reserve monetary policy over time?
-
Match each of the following descriptions, a through e, with one of the following stages of a product life cycle: (1) development, (2) introduction, (3) growth, (4) maturity, or (5) decline. a. Even...
-
Greenwood Company sold $4,000,000 of 7% first-mortgage bonds on October 1, 2003, at $3,479,683 plus accrued interest. The bonds were dated July 1, 2003; interest payable semiannually on January 1 and...
-
It is possible to buy three - month call options and three - month puts on stock Q . Both options have an exercise price of $ 7 6 and both are worth $ 2 6 . If the interest rate is 6 . 7 5 % a year,...
-
A firm relies on R&D to maintain profitability. The firm needs to determine the maximum amount to invest today (or invest in phase I) for its three-phased project. Phase I: Research. (Invest R{)...
-
3 Data Master is a computer software consulting company. Its three major functional areas are computer programming, information systems consulting, and software training. Cynthia Moore, a pricing...
-
Dulles Electric Company buys 1 million tons of coal annually to burn at its power plant. The company wants to invest in a unit train that will haul coal from the mine to the power house, where it is...
-
The Record of TATA Industrial Company Showing the Following data:- Sales Volume in Units : 100 Million Units Selling Price per Unit : $ 3 Variable Cost per Unit : $ 0.90 Total Fixed Cost : $...
-
Calculate the maximum stress in a 5 mm thick steel plate with an 1 8 mm diameter round hole, given h gross = 1 2 cm and the tensile load is 8 . 5 kN . ( Final answer in MPa ) P h gross d
-
Sasha McGowan is an equity analyst at ABI Asset Management. Sasha has been asked by her investment committee to estimate Samsung Electronic's price in 12 months. Sasha has access to the following...
-
Write about a time when tried to convince someone to do something. what reasons and/or evidence did you offer? were you successful in your argument? why or why not? the position you were trying to...
-
Find a unit vector orthogonal to both u and v. u = 3i + 2j - 5k V = + 1k 10
-
Consider the activities undertaken by a medical clinic in your area. Required 1. Do you consider a job order cost accounting system appropriate for the clinic? 2. Identify as many factors as possible...
-
Engineers are testing company fleet vehicle fuel economy (miles per gallon) performance by using different types of fuel. One vehicle of each size is tested. Does this sample provide sufficient...
-
At a University of Colorado woman's home basketball game, a random sample of 25 con- cession purchases showed a mean of $7.12 with a standard deviation of $2.14. For the next week's home game, the...
-
A manufacturing project has five independent phases whose completion must be sequential. The time to complete each phase is a random variable. The mean and standard deviation of the time for each...
-
Barium sulfide is produced by reacting barium sulfate ore with coal. The result is barium black ash, which is \(\mathrm{BaS}\) plus insoluble solids. Since \(\mathrm{BaS}\) is soluble in water, it...
-
Batch leaching is similar to batch extraction, and the equations developed in Section 13.6 can be adapted when the solution is dilute or there is an insoluble solid matrix. We have \(12.5...
-
Repeat Problem 14.D14 except the feed has \(4.5 \mathrm{~kg}\) liquid \(/ \mathrm{kg}\) solids. Problem 14.D14 Barium sulfide is produced by reacting barium sulfate ore with coal. The result is...

Study smarter with the SolutionInn App


