. For each of the following individual molecules check all the descriptions that apply. H3COOCH3 H...
Fantastic news! We've Found the answer you've been seeking!
Question:
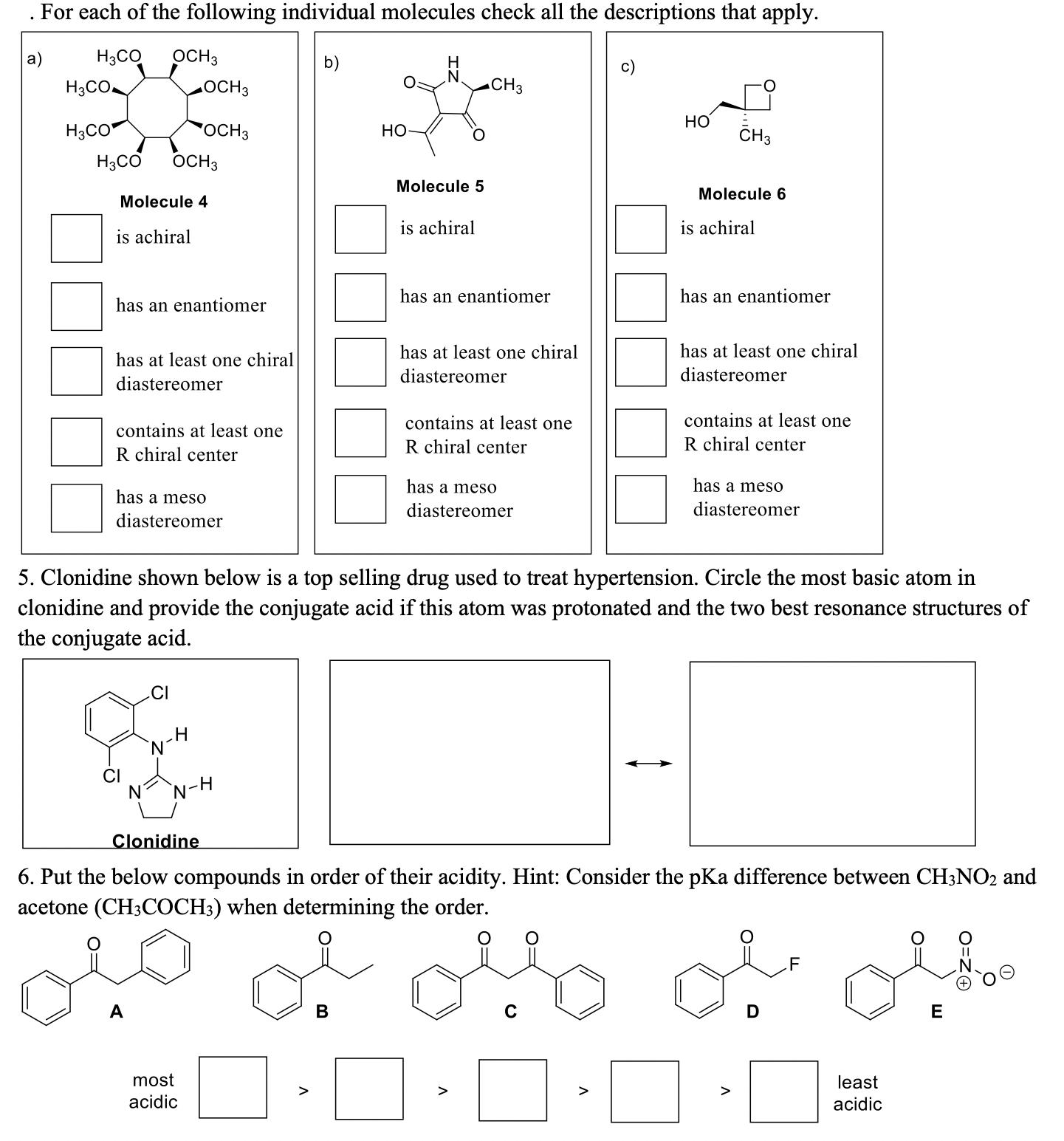
Transcribed Image Text:
. For each of the following individual molecules check all the descriptions that apply. H3COOCH3 H CH3 H3CO OCH 3 H3CO OCH 3 HO H3CO OCH 3 Molecule 4 is achiral Molecule 5 is achiral HO CH3 Molecule 6 is achiral has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomer has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomer has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomer 5. Clonidine shown below is a top selling drug used to treat hypertension. Circle the most basic atom in clonidine and provide the conjugate acid if this atom was protonated and the two best resonance structures of the conjugate acid. CI N H N-H Clonidine 6. Put the below compounds in order of their acidity. Hint: Consider the pKa difference between CH3NO2 and acetone (CH3COCH3) when determining the order. A most acidic B Λ D F E least > acidic . For each of the following individual molecules check all the descriptions that apply. H3COOCH3 H CH3 H3CO OCH 3 H3CO OCH 3 HO H3CO OCH 3 Molecule 4 is achiral Molecule 5 is achiral HO CH3 Molecule 6 is achiral has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomer has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomer has an enantiomer has at least one chiral diastereomer contains at least one R chiral center has a meso diastereomer 5. Clonidine shown below is a top selling drug used to treat hypertension. Circle the most basic atom in clonidine and provide the conjugate acid if this atom was protonated and the two best resonance structures of the conjugate acid. CI N H N-H Clonidine 6. Put the below compounds in order of their acidity. Hint: Consider the pKa difference between CH3NO2 and acetone (CH3COCH3) when determining the order. A most acidic B Λ D F E least > acidic
Expert Answer:
Related Book For 

An Introduction To Statistical Methods And Data Analysis
ISBN: 9781305465527
7th Edition
Authors: R. Lyman Ott, Micheal T. Longnecker
Posted Date:
Students also viewed these programming questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
In the 1D Arrays Unit, you created a command-line Contact Manager v1. Now upgrade the software to include a graphic user interface. Your contact manager must: Have a menu bar that provides access to...
-
QUIZ... Let D be a poset and let f : D D be a monotone function. (i) Give the definition of the least pre-fixed point, fix (f), of f. Show that fix (f) is a fixed point of f. [5 marks] (ii) Show that...
-
Bingl is one the provinces in Turkey with a very high earthquake risk, since the city center is located on the fault and is very close to the East Anatolian Fault Zone, which is also one of the most...
-
A heat engine follows the cycle shown in the figure. (a) How much net work is done by the engine in one cycle? (b) What is the heat flow into the engine per cycle? 4.00 atm 1.00 atm 0.200 m 0.800 m3 T
-
https: / / media 1 . cheggcdn.com / coop / 8 6 9 / 8 6 9 a 2 4 3 9 - d 1 0 3 - 4 c 6 5 - 8 7 1 d - ebc 2 3 b 4 4 0 0 8 e / 1 6 0 3 6 9 5 0 0 0 9 3 8 _ YangInteriors.png ? u = 0 7 5 4 6 bee - adfa - 4...
-
The following financial information is known about Pfizer, Inc. for the year ended December 31, 2015. (all amounts in millions): yom a. The balance of cash and cash equivalents was \($3,641\). b....
-
The inventory of 3T Company on December 31, 2008, consists of these items. Instructions (a) Determine the inventory as of December 31, 2008, by the lower-of-cost-or-market method, applying this...
-
? _ _ _ _ i s a c l a s s o f s o f t w a r e u s e d t o m e e t o r g a n i z a t i o n - w i d e b u s i n e s s n e e d s a n d t y p i c a l l y s h a r e s d a t a w i t h o t h e r e n t e r p...
-
Wayland Custom Woodworking is a firm that manufactures custom cabinets and woodwork for business and residential customers. Students will have the opportunity to establish payroll records and to...
-
What is the HTTP request type which requests status information but not a copy of the document? Get O Head Post O Put O Last-modified O None of the other options
-
Who are the people working at VC firms?
-
When might investors use tranched as opposed to staged financing?
-
What is the option value of abandonment? How can one estimate it?
-
What is the intent behind anti-dilution clauses? Who benefits most from the full-ratchet clause?
-
What are the pros and cons for investors to stage their financial commitments?
-
RAD Co.s most recent free cash flow was $1,000,000. The firm is expected to grow free cash flow at a constant rate of 3% per year forever. RAD Co. has $2,000,000 of debt, $800,000 of preferred stock,...
-
a) Show that (a, b) := {{a}, {b}} does not satisfy the ordered pair axiom. b) Determine whether each of the following statements is true or false. (Give a reason in each case): (i) {a, b} C (a, b)....
-
A group of researchers at a company that produces a leading brand of ice cream design an experiment to evaluate the impact of several artificial sweeteners on the texture of the product. It is well...
-
The baggage limit for an airplane is set at 100 pounds per passenger. Thus, for an airplane with 200 passenger seats, there would be a limit of 20,000 pounds. The weight of the baggage of an...
-
Find the following percentiles for an F distribution with the following specifications: a. = .05, df1 = 14, df2 = 9 b. = .025, df1 = 39, df2 = 27 c. = .01, df1 = 50, df2 = 39 d. = .10, df1 = 39,...
-
Information systems auditors should be most concerned about management's decision to undertake outsourcing of the information systems function when: a. A long-term contract with the outsourcing...
-
Which of the following statements about the implications of privacy legislation for auditors most likely is false? a. As users of personal data, auditors will be exempt from privacy legislation...
-
Which of the following is not an implication of interorganizational information systems and virtual organizations for the conduct of audit work? a. Processing controls become more important than...

Study smarter with the SolutionInn App



