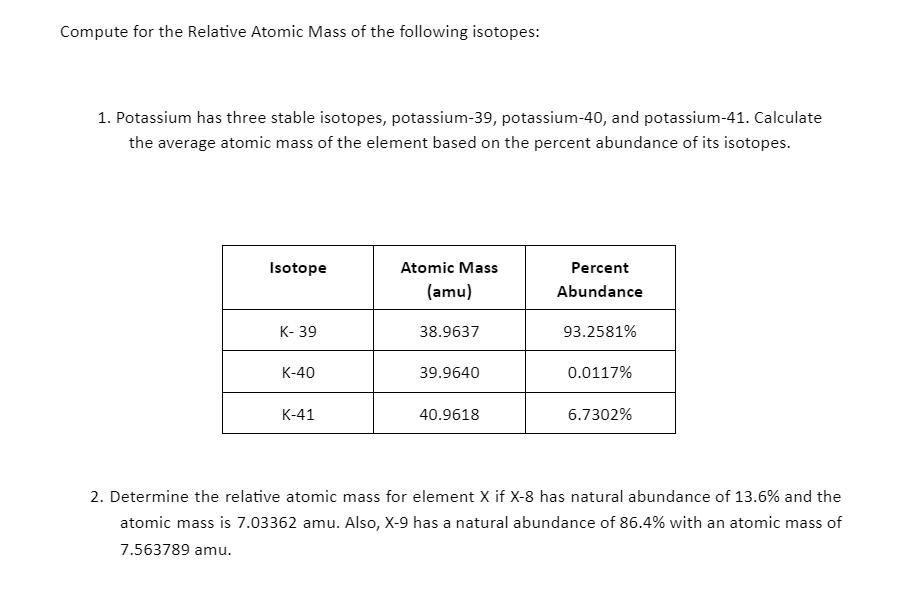
Compute for the Relative Atomic Mass of the following isotopes: 1. Potassium has three stable isotopes,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Compute for the Relative Atomic Mass of the following isotopes: 1. Potassium has three stable isotopes, potassium-39, potassium-40, and potassium-41. Calculate the average atomic mass of the element based on the percent abundance of its isotopes. Isotope K-39 K-40 K-41 Atomic Mass (amu) 38.9637 39.9640 40.9618 Percent Abundance 93.2581% 0.0117% 6.7302% 2. Determine the relative atomic mass for element X if X-8 has natural abundance of 13.6% and the atomic mass is 7.03362 amu. Also, X-9 has a natural abundance of 86.4% with an atomic mass of 7.563789 amu. Compute for the Relative Atomic Mass of the following isotopes: 1. Potassium has three stable isotopes, potassium-39, potassium-40, and potassium-41. Calculate the average atomic mass of the element based on the percent abundance of its isotopes. Isotope K-39 K-40 K-41 Atomic Mass (amu) 38.9637 39.9640 40.9618 Percent Abundance 93.2581% 0.0117% 6.7302% 2. Determine the relative atomic mass for element X if X-8 has natural abundance of 13.6% and the atomic mass is 7.03362 amu. Also, X-9 has a natural abundance of 86.4% with an atomic mass of 7.563789 amu.
Expert Answer:
Answer rating: 100% (QA)
1 The average atomic mass of an element is calculated by taking the weighted average of the masses o... View the full answer

Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Posted Date:
Students also viewed these chemistry questions
-
An element consists of 1.40% of an isotope with mass 203.973 amu, 24.10% of an isotope with mass 205.9745 amu, 22.10% of an isotope with mass 206.9759 amu, and 52.40% of an isotope with mass 207.9766...
-
Calculate the atomic mass of an element with two naturally occurring isotopes, from the following data: What is the identity of element X? Fractional Isotope Isotopic Mass (amu) Abundance X-63 X-65...
-
Determine the atomic mass of ruthenium from the given abundance and mass data Ruthenium-96 Ruthenium-98 Ruthenium-99 Ruthenium-100 Ruthenium-101 Ruthenium-102 Ruthenium-104 5.54% 1.87% 12.76% 12.60%...
-
Which of the following statements are true about REST? Pick ONE OR MORE options Logical URLs should be used instead of physical URLS Adal URLs must always be used in REST response A paging technique...
-
Effect of Stock Dividend Favre Company has a history of paying cash dividends on its common stock. However, the firm did not have a particularly profitable year in 2010. At the end of the year, Favre...
-
Let Ai be the set of all nonempty bit strings (that is, bit strings of length at least one) of length not exceeding i. Find (a) (b) UA
-
Computing unit costs at different levels of production Perfumeria, Inc., budgeted for 12,000 bottles of product Si during the month of May. The unit cost of Si was \(\$ 20\), consisting of direct...
-
The Quick Copy center for document copying is deciding where to locate a new facility. The annual fixed and variable costs for each site it is considering have been estimated as follows: If demand is...
-
A project requires buying a $30,000 equipment, which will be depreciated straight-line to zero over its 5-year life. The project also requires an immediate increase in net working capital of $4,000,...
-
Explain why correctly owning assets is important to the personal finances of people, especially couples.
-
Number of sectors per track increases as we move from inner zone to outer zone, because Select one: a. angular velocity of disk rotation can remain constant. b. the drive can decrease its rotation...
-
Why is financial analysis & Profit planning critical to an operation's optimal success?
-
Estimate the heat of vaporization of diethyl ether at its normal boiling point using Trouton's rule and Chen's rule and compare the results with a tabulated value of this quantity. Calculate the...
-
'Sum' from the book Sum: Forty Tales from the Afterlife by the neuroscientist David Eagleman. 1. What do you think the author is trying to say via this short story? Explain your answer. 2. Do you...
-
Consider the long-run cost curve below. The upward-sloping portion on the right side illustrates Cost (in dollars) ATC Long-Run Cost Curve ATC ATC ATC ATC ATC. 0 150 275 350 450 650 800 900 Quantity...
-
Why do global managers need to understand the debate on globalization?
-
The mathematics department of a college has 5 male professors, 9 female professors, 13 male teaching assistants, and 9 female teaching assistants. If a person is selected at random from the group,...
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Benzene reacts with bromine. a. Write a balanced chemical equation for this reaction. b. Name the catalyst used. c. What visual observations could be made during the reaction? d. Benzene will also...
-
a. Write equilibrium expressions for the solubility products of the following: i. Fe(OH) 2 ii. Fe 2 S 3 iii. Al(OH) 3 b. State the units of solubility product for each of the compounds in part a.
-
a. Explain why a cation with a smaller ionic radius has a higher charge density. b. Which one of the following ions will be the best polariser of the large nitrate ion? Explain your answer. Cs + Li +...
-
Research evidence suggests that, in practice, a cost-plus approach influences many pricing decisions. Required: What is meant by cost-plus pricing and what are the problems of using this approach? E-1
-
Jerry and Co. is a large computer consultancy business that has a division specialising in robotics. Can you identify three non-financial measures that might be used to help assess the performance of...
-
What are the reasons for holding inventories? Are these reasons different from the rea- sons for holding cash? R-3

Study smarter with the SolutionInn App


