Consider a Hydrogen atom in the state n = 2. = 1, m=0 the value ofr...
Fantastic news! We've Found the answer you've been seeking!
Question:
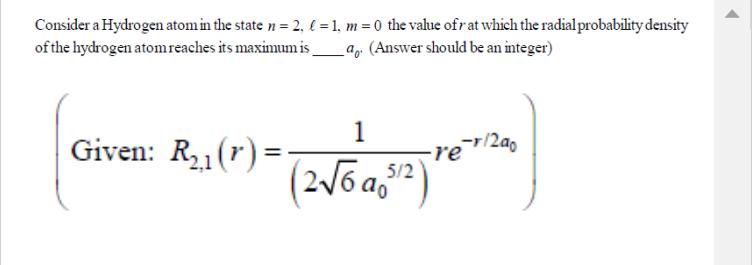
Transcribed Image Text:
Consider a Hydrogen atom in the state n = 2. = 1, m=0 the value ofr at which the radial probability density of the hydrogen atom reaches its maximum is a. (Answer should be an integer) Given: R₂₁(r)= 1 (2√6a5¹²) -re-r/2a0 Consider a Hydrogen atom in the state n = 2. = 1, m=0 the value ofr at which the radial probability density of the hydrogen atom reaches its maximum is a. (Answer should be an integer) Given: R₂₁(r)= 1 (2√6a5¹²) -re-r/2a0
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
Consider a hydrogen atom in the state. (a) For what value of, is the potential energy U(r) equal to the total energy E? Express your answer in terms of a. This value of, is called the classical...
-
A particle collides with a hydrogen atom in the n = 2 state, transferring 15.0 eV of energy to the atom. As a result, the electron breaks away from the hydrogen nucleus. What is the kinetic energy of...
-
A hydrogen atom in a 3p state is placed in a uniform external magnetic field B. Consider the interaction of the magnetic field with the atom's orbital magnetic dipole moment. (a) What field magnitude...
-
Oakey Wines is considering expanding on from the cafe to build a restaurant that will provide an evening experience of a meal, wine and entertainment. The package will be used to showcase new season...
-
A variable-frequency voltage source drives the network in figure. Determine the resonant frequency, Q, BW, and the average power dissipated by the network atresonance. w- 100 n 50 mH 12 cos wf V+ :5 F
-
American Express Travel Related Services ("Amex") sells Amex Travelers Cheques ("TCs"), which are preprinted checks for specified amounts with a unique serial number and no expiration date. Amex is...
-
A popular term for make-or-buy decisions is outsourcing decisions. There are many examples of outsourcing, from Nikes outsourcing of nearly all its production activities to small firms outsourcing of...
-
Suppose that there is technological change that reduces the cost of recruiting for firms. Using the DMP model, determine the effects on the unemployment rate, the vacancy rate, the labor force, the...
-
The following data give the family size (x) and number of pets per family (y) for a random sample of 13 families. 1 6 4 2 2 y 4 9 10 4 23 Create a scatterplot for the data. 11 y 10 9 8- 7 65 6 43 1 2...
-
Complete Tsate's Form 1040-SR, Schedules A, B and D, Form 8949, Form 6252 and Qualified Dividends and Capital Gain Tax Worksheet. Tsate Kongia (birthdate 02/14/1954) is an unmarried high school...
-
the rate of change dp/dt of the number of yeast in a tank is modeled by a logistical differential equation. the maximum capacity of the tank is 794 yeast. at 5pm, the number of yeast in the tank is...
-
(Related to Checkpoint 5.6) (Solving for i) You lend a friend $10,000, for which your friend will repay you $27,027 at the end of 5 years. What interest rate are you charging your "friend"? The...
-
Describe how your department or service delivery would need to be adjusted. For example, would you downsize, bring in more interns ( remember the hidden cost ) , reduce hours, decrease professional...
-
Xenex Inc., a supplier of hospital room disinfection systems, has a cost of capital of 12 percent. To fairly evaluate projects and adjust for risk, it adds or subtracts 2 percentage points to the...
-
Create mind-map or word-web with all of the ways you contribute to your school community. You can create it digitally or by hand. Insert a picture of it below: If a classmate of yours wanted to...
-
Problem 1 (Powers of Two Without Going Over) Write and test a function ced pow_of_two() that takes a positive integer num as the input parameter and returns another integer that is the largest power...
-
Find the present value of an annuity with payments of $600 at the end of every three months for 9 years. The interest rate is 10% compounded quarterly The present value of the annuity is $ (Round the...
-
A condenser (heat exchanger) brings 1 kg/s water flow at 10 kPa quality 95% to saturated liquid at 10 kPa, as shown in Fig. P4.91. The cooling is done by lake water at 20C that returns to the lake at...
-
Consider the condensation of the diamine H2N(CH2)nNH2 with sebacic acid, (HOOC)(CH2)8(COOH), to obtain the polyamide. The polyamide has the following composition: 67.2% C
-
Sodium carbonate, Na2CO3, can be prepared by heating sodium hydrogen carbonate, NaHCO3. 2NaHCO3(s) Na2CO3(s) + H2O(g) + CO2(g) Estimate the temperature at which NaHCO3 decomposes to products at 1...
-
A white crystalline material contained sodium acetate and sodium chloride. To determine the composition of this mixture, a researcher dissolved 0.613 g of this mixture in water to give 25.0 mL of...
-
What sort of cash organisation is generally in place for highly decentralised groups?
-
Is the risk of bankruptcy of a subsidiary an obstacle to cash pooling for a group which balances its accounts daily?
-
What is the main argument against full cash pooling for a group?

Study smarter with the SolutionInn App



