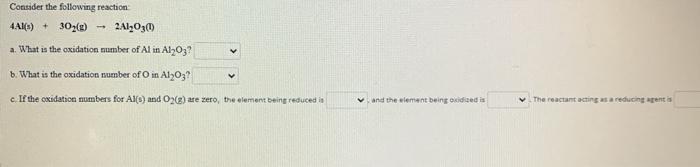
Consider the following reaction: 4.Al(s) + 302(g) 1 2AlO3(1) a. What is the oxidation number of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Consider the following reaction: 4.Al(s) + 302(g) 1 2Al₂O3(1) a. What is the oxidation number of Al in Al2O3? b. What is the oxidation number of O in Al₂O37 c. If the oxidation numbers for Al(s) and O2(g) are zero, the element being reduced is and the element being oxidized is The reactant acting as a reducing agent is Consider the following reaction: 4.Al(s) + 302(g) 1 2Al₂O3(1) a. What is the oxidation number of Al in Al2O3? b. What is the oxidation number of O in Al₂O37 c. If the oxidation numbers for Al(s) and O2(g) are zero, the element being reduced is and the element being oxidized is The reactant acting as a reducing agent is
Expert Answer:
Answer rating: 100% (QA)
Sol 4 Als 3 O g 2 AlO3 1 a Oxidation number of oxygen is generally t... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemical engineering questions
-
Consider the following reaction at some temperature: H2O(g) + CO(g) H2(g) + CO2(g) K = 2.0 Some molecules of H2O and CO are placed in a 1.0- L container as shown below. When equilibrium is reached,...
-
Consider the following reaction at 800. K: N2(g) + 3F2(g) 2NF3(g) An equilibrium mixture contains the following partial pressures: PN2 = 0.021 atm, PF2 = 0.063 atm, and PNF3 = 0.48 atm. Calculate Go...
-
Consider the following reaction at 298 K: 2SO2(g) + O2(g) 2SO3(g) An equilibrium mixture contains O2(g) and SO3(g) at partial pressures of 0.50 atm and 2.0 atm, respectively. Using data from...
-
The accountant for a subunit of Mountain Sports Company went on vacation before completing the subunits monthly performance report. This is as far as she got: Requirements 1. Complete the performance...
-
1. What business research problem does Royal Barton face? What are his information needs? Outline some survey research objectives for a research project on the Royal Bee system. 2. What type of...
-
Presented below are the financial statements of Kurtzel Company. Additional data: 1. Depreciation expense was $17,500. 2. Dividends declared and paid were $20,000. 3. During the year equipment was...
-
8. Ryder leased a truck to Florida Food Service; Powers, an employee, drove it to make deliveries. He noticed that the door strap used to close the rear door was frayed, and he asked Ryder to fix it....
-
The petty cash fund of Fonzarellis Auto Repair Service, a sole proprietorship, contains the following. The general ledger account Petty Cash has a balance of $300. Instructions Prepare the journal...
-
Zoe Corporation has the following information for the month of March: Cost of direct materials used in production $16,431 Direct labor 26,000 Factory overhead 39,890 Work in process inventory, March...
-
A camera is listed for $599 less 30%, 20%, and 5%. (a) What is the net price? (b) What is the total amount of discount allowed? (c) What is the exact single rate of discount that was allowed?
-
Question 1 For the year 2018, the gross profit of Alpha Company was $80,000; the cost of goods manufactured was $400,000; the beginning inventories of goods in process and finished goods were $28,000...
-
Distinguish between embezzlement and robbery.
-
May internal auditors consider their responsibilities for the prevention and detection of fraud discharged if they find written procedures which provide satisfactory controls designed to eliminate...
-
An internal auditor tells man^ement: "The courts have held that an auditor cannot be regarded as an insurer against fraud. Ergo, I can take no responsibility for its prevention or detection in this...
-
What two forms of pressure can cause managers to defraud their companies?
-
Leadership, properly carried out, can best be described as: a. Carefully following the managerial functions. b. Achieving objectives. c. Demonstrating concern for the needs of subordinates. d....
-
Answer the following prompts Make a list of ten groups you are or have been a member of (e.g. classes, teams, youth groups, workplaces/professional roles) Choose two that have been really important...
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
The following electron configurations represent excited states. Identify the element, and write its ground-state condensed electron configuration. (a) 1s2 2s2 3p2 4p1, (b) [Ar] 3d10 4s1 4p4 5s1, (c)...
-
Many gases are shipped in high-pressure containers. Consider a steel tank whose volume is 55.0 gallons that contains O2 gas at a pressure of 16,500 kPa at 23oC. (a) What mass of O2 does the tank...
-
KCl has the same structure as NaCl. The length of the unit cell is 6.28 . The density of KCl is 1.984 g/cm3, and its formula mass is 74.55 amu. Using this information, calculate Avogadro's number?
-
Rowe Corporation authorized \(\$ 5,000\) of \(8 \%\) (cash interest payable semiannually) 10 -year bonds. The bonds were dated January 1, 2020; interest dates are June 30 and December 31. Assume...
-
For each of the following debt scenarios described below, identify the bond type that applies. a. Bonds issued by a public corporation b. Mortgage note issued for commercial property c. Bonds issued...
-
An eight-year, \(6 \%, \$ 1,000\) bond (cash interest payable annually) is sold to yield \(6 \%\) interest. Compute the bond selling price.

Study smarter with the SolutionInn App


