DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
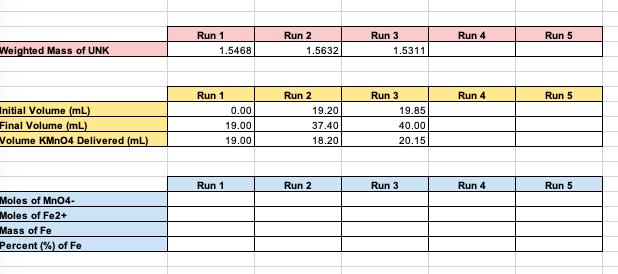
DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your professor and record the unknown number in your notebook. Weigh out 3-4 samples of between 1.5000g and 2.0000g ferrous ammonium sulfate unknown and place in 250 mL flasks. Add 15 mL of distilled water and 1 mL of concentrated HCI to each sample. Swirl to dissolve sample completely. Gently warm on a hot plate at your bench. Wash down the sides of the flask with some distilled water. Add 0.2 M Potassium Permanganate solution (not your standardized solution) dropwise until a distinct yellow color forms. (should be about 2 drops) Carry out the following procedure individually on each sample. Add 25 mL of the Zimmermann-Reinhardt reagent and 300 mL of distilled water. Titrate immediately with your standardized KMnO4 solution to the first faint pink color that persists for 15 to 30 seconds. Do not titrate rapidly at any time. The pink color results from excess MnO4, indicating that all Fe²+ has been oxidized to Fe³+, Weighted Mass of UNK Initial Volume (mL) Final Volume (mL) Volume KMnO4 Delivered (mL) Moles of MnO4- Moles of Fe2+ Mass of Fe Percent (%) of Fe Run 1 1.5468 0.00 19.00 19.00 Run 1 Run 1 Run 2 1.5632 19.20 37.40 18.20 Run 2 Run 2 Run 3 1.5311 19.85 40.00 20.15 Run 3 Run 3 Run 4 Run 4 Run 4 Run 5 Run 5 Run 5 DETERMINATION OF IRON CONTENT IN A FERROUS AMMONIUM SULFATE UNKNOWN Obtain an unknown sample from your professor and record the unknown number in your notebook. Weigh out 3-4 samples of between 1.5000g and 2.0000g ferrous ammonium sulfate unknown and place in 250 mL flasks. Add 15 mL of distilled water and 1 mL of concentrated HCI to each sample. Swirl to dissolve sample completely. Gently warm on a hot plate at your bench. Wash down the sides of the flask with some distilled water. Add 0.2 M Potassium Permanganate solution (not your standardized solution) dropwise until a distinct yellow color forms. (should be about 2 drops) Carry out the following procedure individually on each sample. Add 25 mL of the Zimmermann-Reinhardt reagent and 300 mL of distilled water. Titrate immediately with your standardized KMnO4 solution to the first faint pink color that persists for 15 to 30 seconds. Do not titrate rapidly at any time. The pink color results from excess MnO4, indicating that all Fe²+ has been oxidized to Fe³+, Weighted Mass of UNK Initial Volume (mL) Final Volume (mL) Volume KMnO4 Delivered (mL) Moles of MnO4- Moles of Fe2+ Mass of Fe Percent (%) of Fe Run 1 1.5468 0.00 19.00 19.00 Run 1 Run 1 Run 2 1.5632 19.20 37.40 18.20 Run 2 Run 2 Run 3 1.5311 19.85 40.00 20.15 Run 3 Run 3 Run 4 Run 4 Run 4 Run 5 Run 5 Run 5
Expert Answer:
Answer rating: 100% (QA)
Molasity of Initial V ml 010 1920 1985 Final V ml 190 3740 4000 V 190 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these mechanical engineering questions
-
A dilute solution of potassium permanganate in water at 25C was prepared. The solution was in a horizontal tube of length 10 cm, and at first there was a linear gradation of intensity of the purple...
-
Potassium sulfate solution reacts with barium bromide solution to produce a precipitate of barium sulfate and a solution of potassium bromide. Write the molecular equation for this reaction. Then...
-
A 35.2-mL, 1.66 M KMnO4 solution is mixed with 16.7mL of 0.892 M KMnO4 solution. Calculate the concentration of the final solution.
-
Susan recently quit working for a local firm and has yet to find a new job. She knows she can maintain her health insurance from her old employer due to COBRA. How much will it likely cost her for...
-
A bond has a 7 percent coupon rate, a face value of $1,000, and a maturity of four years. On a time line, lay out the cash flows for the bond.
-
6x 8y = 16 Determine the slope and the y-intercept. Use the slope and the y-intercept to graph the equation by hand.
-
compare and contrast models of learning,
-
1. Who would you recommend Stacey Thomas select to serve as project manager? Why? 2. How would you recommend this project be organized? Functional project? Pure project? Matrix? Why? 3. Do you agree...
-
\f
-
Consider the following income tax footnote information for Oracle for the fiscal year ended May 31, 2019 (fiscal year 2019). The following is a geographical breakdown of income before the provision...
-
Lubricants, Incorporated, produces a special kind of grease widely used by race car drivers. The grease is produced in two processing departments-Refining and Blending. Raw materials are Introduced...
-
In light of theconsequences of being uninsured (i.e., higher overall out-of-pocket health costs, or people not getting healthcare services when needed), what else should government do to meet the...
-
What is the difference between MS Access and enterprise-wide databases such as MySQL and Oracle? Also, research the new generation of the database called NoSQL database and explain its advantages...
-
The Company is expected to announce their annual dividend tomorrow. One year ago they paid a dividend of $2.30 , and 4 years ago they paid $1.85 .You believe that future dividends will grow by the...
-
Please help me in creating the following two functions: A function called prnDoubles that prints all elements of a double array, comma-separated with one digit after the decimal point. an another...
-
AT&T Wireless: Ma Bell Becomes Ma Again on pp. 762-765 in the textbook. Assume that you are a data analyst for AT&T who has been tasked with analysing the survey data. Refer to the accompanying JMP...
-
Capital Budgeting A company is evaluating two investment opportunities: Project C: Initial investment: $100,000 Yearly Cash Inflows: $25,000 for 6 years Project D: Initial investment: $150,000 Yearly...
-
Let (X. A. p) be a measure space. Show that for any A,B A, we have the equality: (AUB)+(An B) = (A) + (B).
-
An acidic solution containing 0.010 M La3+ is treated with NaOH until La(OH)3 precipitates. At what pH does this occur?
-
Sulfide ion was determined by indirect titration with EDTA. To a solution containing 25.00 mL of 0.04332 M Cu(ClO4)2 plus 15 mL of 1M acetate buffer (pH 4.5) were added 25.00 mL of unknown sulfide...
-
What are the general steps in developing an isocratic separation for reversed-phase chromatography with one organic solvent and temperature as variables?
-
Selected transactions for Neve Campbell Company during its first month in business are presented below. Sept. 1 Invested $10,000 cash in the business in exchange for common stock. 5 Purchased...
-
Food To Go is a local catering service. Conceptually, when should Food To Go recognize revenue from its catering service? a. At the date the invoice is mailed to the customer b. At the date the...
-
The accounts in the ledger of Speedy Delivery Service contain the following balances on July 31, 2006. Instructions Prepare a trial balance with the accounts arranged as illustrated in the chapter...

Study smarter with the SolutionInn App


