Express the results of the following calculations with the correct number of significant figures. 3.01-0.23 0.235...
Fantastic news! We've Found the answer you've been seeking!
Question:
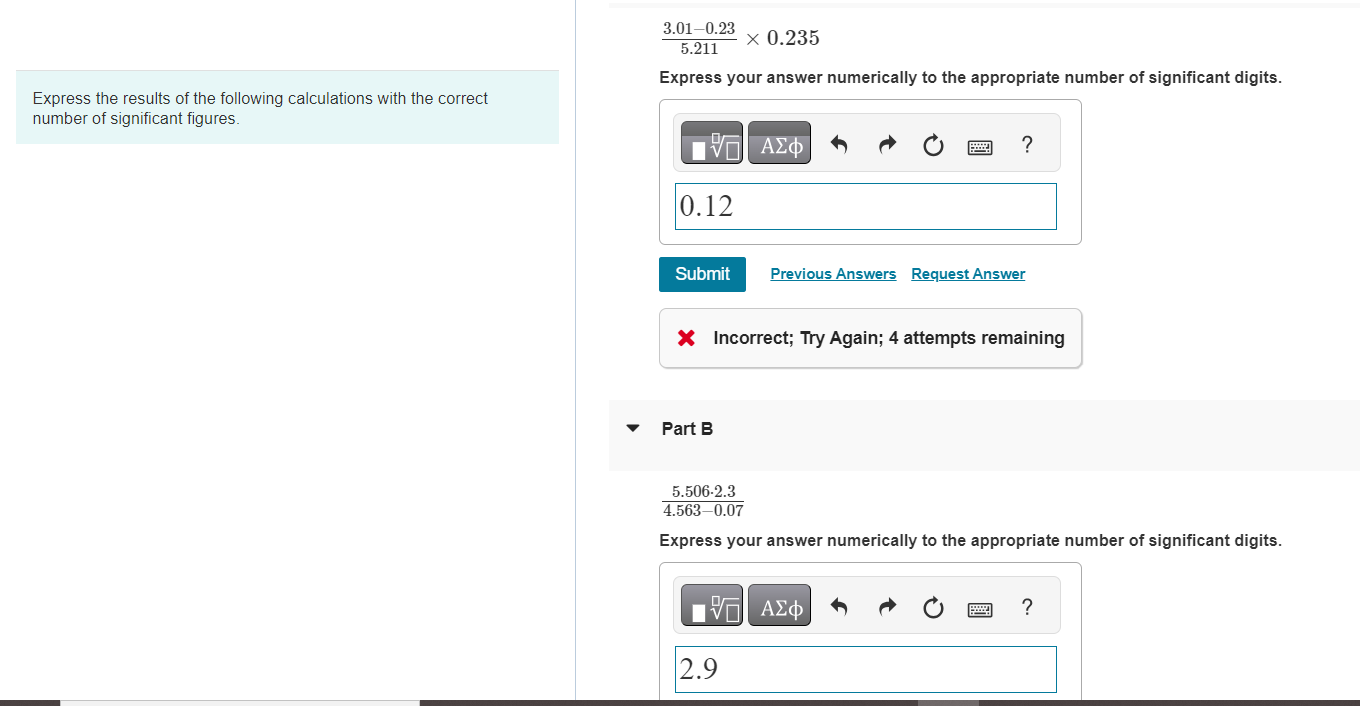
Transcribed Image Text:
Express the results of the following calculations with the correct number of significant figures. 3.01-0.23 0.235 5.211 Express your answer numerically to the appropriate number of significant digits. || 0.12 Submit Part B X Incorrect; Try Again; 4 attempts remaining ? Previous Answers Request Answer 5.506-2.3 4.563-0.07 Express your answer numerically to the appropriate number of significant digits. VE 2.9 ? Express the results of the following calculations with the correct number of significant figures. 3.01-0.23 0.235 5.211 Express your answer numerically to the appropriate number of significant digits. || 0.12 Submit Part B X Incorrect; Try Again; 4 attempts remaining ? Previous Answers Request Answer 5.506-2.3 4.563-0.07 Express your answer numerically to the appropriate number of significant digits. VE 2.9 ?
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Use MYSTAT or LazStats to reproduce the analysis in Exercise 16.23. The data follow. 34 65 5 3 2 5 2 4 6 8 75 61 2 Am 48 29 34 6 8 6 9 54 8 7 60 5 5 25 49 4 5 239 20 5 3 2 7 4 30 4 4 4 9 9
-
If a 2 Ã 2 matrix is multiplied on the left by the given matrices, what elementary row operation is performed on that matrix? (a) (b) (c) 0 01 9
-
Who makes up the crisis team? Develop a CMT for the organization you're creating the CCP for. What organizational units should be represented on the team (e.g., public relations, IT, etc.)? Tailor...
-
A 2-kg picture is hung by two wires of equal length. Each makes an angle of ? with the horizontal, as shown in Figure. (a) Find the general equation for the tension T, given ? and weight w for the...
-
A reservoir has the storage-discharge relationship below. Route the inflow hydrograph in problem 4.16 through the reservoir, assuming an initial storage of 52 106 m3 of water. In problem 4.16 Time...
-
Describe Gantt charts and how they are useful in job shop scheduling.
-
Aging Method Bad Debt Expense} Cindy Bagnal, the manager of Cayce Printing Service, has provided you with the following aging schedule for Cayce's accounts receivable: Cindy indicates that the \(\$...
-
On January 1, 2010, Metco, Inc., had issued an outstanding 574,600 shares of $2 par value common stock . On March 15, 2010, Metco, Inc., purchased for its treasury 4,400 shares of its common stock at...
-
Murray s So - Soft - Sheets were becoming incredibly popular. Murray Company initially planned to produce and sell the sheets, but then decided to add another layer of production: using the very soft...
-
Calculate the following four reliability coefficients using the Pearson product-moment correlation formula, correlate the scores to determine the reliability coefficient (xx). Show your work. 3....
-
The following information comes from the 2021 Annual Report to stockholders of Composition Inc. (in thousands): From the Statement of Changes in Shareholders' Equity: BALANCES AT December 31, 2019...
-
What is the physical principle that is used to separate gases and liquids? For a liquid droplet of 9 microns, determine the settling velocity in a vertical separator where the gas is also present....
-
Undertake Threat of new entrants (Porter's 5 Forces analysis) to determine how much profit potential exists in the industry in which your selected business operates. If you select a conglomerate...
-
OSU TECS y! y! y! y! y! Y! mpr y! npr y! W y! y! y! y! production-gradescope-uploads.s3-us-west-2.amazonaws.com/uploads/pdf_attachment/file/95736765/WHW5W23.pdf?X-Amz-Algorithm=AWS4-HMA... C...
-
The metal niobium melts at a temperature of 2468 C and boils at 4742 C, whereas the metal gallium melts at a temperature of 30 C and boils at 2403 C. (a) Which metal will be more volatile at room...
-
11. Consider the following problem description: y vods adinol nolllzogm motavador (a) List and provide rationale for three design goals for the above system. Briefly discuss if any two of them are...
-
The summarised financial statements for three individual companies at 31 December 2020 are: Statements of financial position as at 31 December 2020 Assets Sun Earth Pluto Non-current assets I Land...
-
The National Collegiate Athletic Association (NCAA) and the National Federation of State High School Associations (NFHS) set a new standard for non-wood baseball bats. Their goal was to ensure that...
-
A reaction has a G = 10.0 kJ and a H = 20.0 kJ. If S = 1.82 10 3 J/K, what was the temperature at which the reaction occurred? a. 6.04 K b. 6.04 10 3 K c. 5.49 10 3 K d. 5.49 K e. 8.55 K
-
You carry out the following reaction by introducing N 2 O 4 into an evacuated flask and observing the concentration change of the product over time. N 2 O 4 (g) 2NO 2 (g) Which one of the curves...
-
Write molecular and net ionic equations for the successive neutralizations of each acidic hydrogen of phosphoric acid by calcium hydroxide solution. Ca3(PO4)2 is insoluble; assume that the acid salts...
-
Amesbury plc produces and distributes computer-controlled machinery. As accountant for the company, you have been provided with the following information regarding the company's activities in...
-
You are the management accountant of Short plc. On 1 October 1993 Short plc issued 10 million \( 1\) preference shares at par, incurring issue costs of \( 100000\). The dividend payable on the...
-
Your managing director has recently read an article which referred to Financial Reporting Standard 4 (FRS 4) - Capital instruments. He has requested a report from you about FRS 4....

Study smarter with the SolutionInn App


