For compounds A and B shown below, provide the reaction conditions needed to remove the protecting...
Fantastic news! We've Found the answer you've been seeking!
Question:
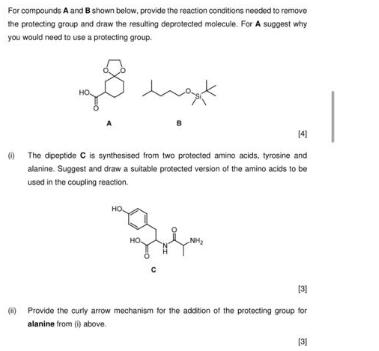
Transcribed Image Text:
For compounds A and B shown below, provide the reaction conditions needed to remove the protecting group and draw the resulting deprotected molecule. For A suggest why you would need to use a protecting group. want 141 (The dipeptide C is synthesised from two protected amino acids, tyrosine and alanine. Suggest and draw a suitable protected version of the amino acids to be used in the coupling reaction. HO HO NH₂ [3] Provide the curly arrow mechanism for the addition of the protecting group for alanine from () above (31 For compounds A and B shown below, provide the reaction conditions needed to remove the protecting group and draw the resulting deprotected molecule. For A suggest why you would need to use a protecting group. want 141 (The dipeptide C is synthesised from two protected amino acids, tyrosine and alanine. Suggest and draw a suitable protected version of the amino acids to be used in the coupling reaction. HO HO NH₂ [3] Provide the curly arrow mechanism for the addition of the protecting group for alanine from () above (31
Expert Answer:
Answer rating: 100% (QA)
Step 12 a For compound A a protecting group is needed because the amino group is a nucleophile and c... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Two isomeric compounds A and B with molecular formula C6H8 decolourize bromine solution in CCl4 and also alkaline KMn04. On catalytic hydrogenation, both yield cyclohexane (C6H12). A shows an...
-
Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra. Compound A: 1.3 ppm (3H, triplet); 3.6 ppm (2H, quartet); 4.1 ppm (2H,...
-
Compounds A and B are carboxylic acids. Identify each one on the basis of its 1H NMR spectrum. (a) Compound A (C3H5ClO2) (Figure 19.10). (b) Compound B (C9H9NO4) has a nitro group attached to an...
-
What is the main difference between n-type and p-type semiconductors?
-
Gamma Medicals stock trades at $90 a share. The company is contemplating a 3-for-2 stock split. Assuming the stock split will have no effect on the total market value of its equity, what will be the...
-
Solve subject to the initial condition T(x, 0) = 1 and the boundary conditions T(0, t) = 0 and T(1, t) = 0. Report your answer in terms of the non-zero Fourier modes. aT at || 2
-
Explain the difference between primary and secondary markets.
-
Yemi Ltd. is a retailer operating in Edmonton, Alberta. Yemi uses the perpetual inventory method. All sales returns from customers result in the goods being returned to inventory; the inventory is...
-
What is one intended outcome of the problem Solving workshop forming working groups to better understand your issues from The PI developing solution to organize problems, that blocked or slowed work...
-
Compare the forward quotes, money market hedges and options with each other to determine the best exchange rate hedge for France (Complete Table 5 on the separate answer sheet) Calculate the exchange...
-
On its most recent financial statements, Gevelyn Inc. reported net sales of $ 2 7 3 , 0 0 0 , accounts receivable of $ 7 7 , 0 0 0 , inventories of $ 6 4 , 0 0 0 , and accounts payable of $ 4 2 , 0 0...
-
In what ways you can develop procedures for emergency extraction of a protectee from a dangerous location or situation. When it comes to the protection agency what is the mission and how can they use...
-
a.) b.) int func5(int A[], int n) { } if (n100) return(0); x = A[1];) for (i = 0; i < sqrt(n); i++) { for (j = 0; j
-
Required: 1) Enter the opening balances from the August 2021 balance sheet into the general ledger accounts. Prepare journal entries for the month of September and post them to the appropriate...
-
The project I will be talking about was a marketing initiative to gain more customers for our checking and savings accounts at a local bank I worked at. There was several of us that made up the team,...
-
4. a) Consider the following set of facts: 1. X is a student. 2. X studies in CSE department. 3. X has passed HSC in the year 2016. 4. Passing year is session. 5. The students of 2016 session of CSE...
-
SCENARIO: The Rocky Ravine Hospital's Maintenance Facility, this facility is responsible for the maintenance of the embedded technology in magnetic resonance imaging (MRI) machines within the...
-
An 8.0 kg crate is pulled 5.0 m up a 30 incline by a rope angled 18 above the incline. The tension in the rope is 120 N, and the crates coefficient of kinetic friction on the incline is 0.25. a. How...
-
Methamphetamine is a notorious street drug. One synthesis involves reductive amination of benzyl methyl ketone with methylamine. What is the structure of methamphetamine?
-
Many carboxylic acids are much better known by their common names than by their systematic names. Some of these follow. Provide a structural formula for each one on the basis of its systematic name....
-
The following are the more stable anomers of the pyranose forms of D-glucose, D-mannose, and D-galactose: HO HOCH2 HOCH, OH CH OH HO HO HO OH HO HO B-D-Glucopyranose 64% at equilibrium)...
-
A packed tower is used to absorb ammonia from air using aqueous sulfuric acid. Gas enters the tower at \(31.0 \mathrm{lbmol} /\left(\mathrm{h}-\mathrm{ft}^{2} ight)\) and is 1.0 \(\mathrm{mol} \%\)...
-
Water originally saturated with carbon tetrachloride \(\left(\mathrm{CCl}_{4} ight)\) at \(25.0^{\circ} \mathrm{C}\) and \(1.0 \mathrm{~atm}\) is stripped with pure air at \(25.0^{\circ} \mathrm{C}\)...
-
We are separating methanol and water in a staged distillation column at total reflux to determine Murphree efficiency. Pressure is \(101.3 \mathrm{kPa}\). The column has a 2.0 -in. head of liquid on...

Study smarter with the SolutionInn App


