How much work is done when 330 reaction at 295. K? 2CH (1) + 150 (g)...
Fantastic news! We've Found the answer you've been seeking!
Question:
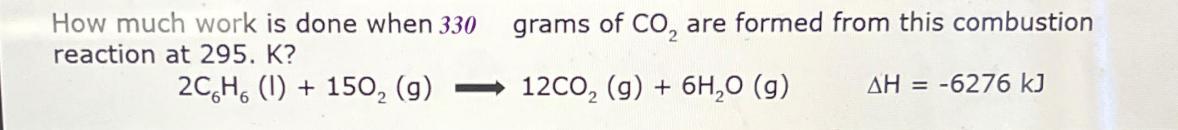
Transcribed Image Text:
How much work is done when 330 reaction at 295. K? 2CH (1) + 150 (g) grams of CO are formed from this combustion 12CO (g) + 6HO (g) AH = -6276 kJ How much work is done when 330 reaction at 295. K? 2CH (1) + 150 (g) grams of CO are formed from this combustion 12CO (g) + 6HO (g) AH = -6276 kJ
Expert Answer:
Answer rating: 100% (QA)
The question is asking about the amount of work done during a combustion reaction that produces 330 grams of CO carbon dioxide The given reaction is t... View the full answer

Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Posted Date:
Students also viewed these chemical engineering questions
-
Case Study Eagersaver.com Eagersaver.com was established in 2005 by the CEO Colette Bevan as an online comparison site primarily focused on car insurance and related products. Since then it has...
-
(1 point) The differential equation -y" = f(t), y(2) = 0. y(5) = 0 discretized with step-size h = has the following impulse responses. %3D 3. 3 y(1) y(2) 4 y(3) 25 2 y(4) 25 4 25 6 25 3 What is the...
-
7. (15 Points) A streaming service charges one fee ($500) for a 2-year subscription. If a 12% nominal annual interest rate with compounding every month is used. a. What is the effective annual...
-
Regulation imperial soccer fields have a length of 91 meters and a width of 39 meters. Show that during a friendly match there are always two players that are not farther from each other than 19...
-
Suppose that c 0 for x [c, d] and (x) = 0 elsewhere in [a, b], prove that R[a, b] and that Rb = a(d - c). [Given > 0 let := /4a and show that if a(d - c + 2).]
-
Is risk reduction in the firm's portfolio of undertakings likely to be best achieved through horizontal integration, vertical integration, or conglomerate-type acquisitions?
-
Which factors increase your stress level?
-
A firm??s product sells for $2 per unit in a highly competitive market. The firm produces output using capital (which it rents at $75 per hour) and labor (which is paid a wage of $15 per hour under a...
-
1.A Japanese bank expects to lend 10 million to a Spanish firm. If negotiations are successful, the loan will be made one month from today. The terms of the loan have already been established: the...
-
Determine the breakeven volume of injections for 2016 using the following formula for the contribution margin ratio approach: Breakeven revenue = Total fixed costs + [(Total variable costs / Total...
-
Whispering Company has two classes of capital stock outstanding: 7%, $20 par preferred and $5 par common. At December 31, 2020, the following accounts were included in stockholders' equity. Preferred...
-
Diffusionless Martensitic Transformation (10P) a. Write at least six important features associated with martensitic transformation. (3p) b. What are the differences between lath and plate martensite...
-
Consider two parallel wires 3 [m] apart. They are oppositely charged. If the electric field strength halfway between them is E = 1.5 104 [V/m], how much excess positive charge rests on a 1 [km]...
-
ADRIAN and BLAINE were enthusiastic hobbyists in metal detecting. From time to time they went to sites together and split the costs of transport and accommodation. They did not operate in partnership...
-
How does the integration of advanced computational methods, like finite element analysis, impact the safety and efficiency of large-scale structural design ?
-
Can you elaborate on the concept of heat capacity, particularly in the context of its dependence on temperature and its role in characterizing the response of a system to changes in thermal energy ?
-
On April 1, Jiro Nozomi created a new travel agency, Adventure Travel. The following transactions occurred during the company's first month. April 1 Nozomi invested $30,000 cash and computer...
-
In 1995 Miguel purchased a home for $130,000. In 2000 he sold it for $170,000 and immediately purchased another one for $180,000, which he sold in 2007 for $235,000. How much taxable capital gain, if...
-
In your own words, define the following symbols or terms: (a) E; (b) F; (c) Anode; (d) Cathode.
-
A chemistry handbook lists the following equation for the vapor pressure of NH 3 (l) as a function of temperature. What is the normal boiling point of NH 3 (l)? log10 P(mmHg) = 9.95028 0.003863T...
-
Ultimately, f G values must be based on experimental results; in many cases, these experimental results are themselves obtained from E values. Early in the twentieth century, g. N. Lewis conceived...
-
Adelphi Video Store maintains the following policies with regard to purchases of new videotapes at each of its branch stores: 1. Employees are required to take vacations, and the duties of employees...
-
At the end of the fiscal year, August 31, 20x6, selected accounts from the adjusted trial balance for Mikhails Delivery, Inc., appeared as follows: Required 1. Using the information given, prepare an...
-
Using the relevant data in E 10, give the entries in T-account form to record each of the transactions under the periodic inventory system. Use of Accounting Records in Internal Control

Study smarter with the SolutionInn App


