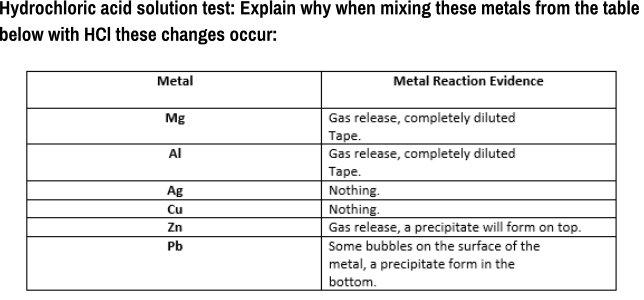
Hydrochloric acid solution test: Explain why when mixing these metals from the table below with HCI...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Hydrochloric acid solution test: Explain why when mixing these metals from the table below with HCI these changes occur: Metal Metal Reaction Evidence Gas release, completely diluted Таре. Gas release, completely diluted Mg Al Таре. Nothing. Ag Cu Nothing. Gas release, a precipitate will form on top. Zn Pb Some bubbles on the surface of the metal, a precipitate form in the bottom. Hydrochloric acid solution test: Explain why when mixing these metals from the table below with HCI these changes occur: Metal Metal Reaction Evidence Gas release, completely diluted Таре. Gas release, completely diluted Mg Al Таре. Nothing. Ag Cu Nothing. Gas release, a precipitate will form on top. Zn Pb Some bubbles on the surface of the metal, a precipitate form in the bottom.
Expert Answer:
Answer rating: 100% (QA)
Reaction of hydrochloric acid with magnesiumMg When hydrochloric acid reacts with magnesium ribbon magnesium chloride is produced and hydrogen gas is ... View the full answer

Posted Date:
Students also viewed these chemical engineering questions
-
the table below with the fraud type(s) listed with the table Do this as a team Note for the Potential Controls section of the table, you will put yourself in the position of a manager of an...
-
From the table below place correctly the phase name, symbol, phase defined and opposite phase. Phase Name AH symbol Phase defined Opposite Enthalpy of fusion AHvap Sublimation solidification Enthalpy...
-
Define briefly what Marx means by inorganic capital? What Marx says about the share of inorganic capital in the long term and what is the consequence of that change on capitalist profits in long term?
-
.How dependent do you think the success of zero-based budgeting would be on the types of attribution models used? How might you increase the odds of success in this budgeting approach? .Advertising,...
-
Interim financial reports are subject to limitations and distortions. Identify and discuss at least two reasons for this.
-
Prove the complex convolution relation given by(3.2.22). 2 x1(n)x;(n): %3D ap v-'dv
-
How can individuals use the present value of an annuity to plan for retirement?
-
Fill in the partially completed inventory record shownhere. Item: AB500 Lot Size Rule: POQ Lead Time: 2 weeks Parent: None Children: AB501, AB511, 521 3 Gross Requirements Schedtuled Receipts: 150...
-
Function f is defined by f(x)=3x+c. If f(6) = 1, what is the value of f(c)? (A) -20 (B) -8 (C) 4 (D) 12
-
Beth and Kate are in partnership. Their financial year ends on 31 December. On 1 January 205 their capitals were: Beth $90,000 and Kate $70,000. The uncompleted current accounts for the year ended 31...
-
Describe how each of the following encapsulated objects allows the user to focus on what the object does with little or no concern for how it does it: How can the C preprocessor determine whether a...
-
What strategies can leaders employ to ensure meaningful engagement with a diverse array of stakeholders, including employees, customers, investors, and the community ?
-
When an individual goes from living in poverty to a middle-class lifestyle, there is clearly a strong relationship between increased wealth and increased happiness. However, once individuals move...
-
With the following information for Aspen Resort, please prepare a contribution margin income statement if 50 rooms are rented in March. Make sure to fill in all the blanks and indicate if the ending...
-
Cryptocurrencies are often seen as an investment opportunity, particularly in countries susceptible to deprecating and unstable national currencies. Oil-rich Nigeria should be one of Africa's...
-
This past year, Abe earned $30,000; Betty earned $150,000; and Candace earned $12,000 (all wages listed are before tax). Betty paid $45,000 in income taxes. Abe paid $7,500 in income taxes and...
-
Review the articles on the BP Gulf Oil spill and corporate social responsibility in Required Readings. Explain how the philosophy of Milton Friedman could have played a role in BP managements...
-
The landing gear of an aircraft with: mass of 2000 kg the spring-mass-damper system Consider that the runway surface is y(t) = 0.2 cos 157.08t stiffness of the spring is 5 x 105 N/m. What is the...
-
After factory overhead has been applied to work in process, what does a credit balance in Factory Overhead indicate?
-
Explain the difference between actual factory overhead and applied factory overhead.
-
To which work sheet column is the debit balance of Cost of Goods Sold extended?

Study smarter with the SolutionInn App

